Wuwei Heavy Ion Center: Heavy Ion Therapy for Stage III NSCLC – A 165-Case Analysis
Wuwei Heavy Ion Center: Clinical Analysis of 165 Cases of Stage III Non-Small Cell Lung Cancer Treated with Heavy Ion (Carbon) Therapy
On March 13, 2025, we reported a clinical analysis of 538 cases of non-small cell lung cancer treated with heavy ion (carbon) therapy at the Wuwei Heavy Ion Center over the past eight years (https://www.wwzlz.cn/news/1956). Today, we provide a further analysis of the treatment outcomes for patients with stage III non-small cell lung cancer from that cohort.
Since treating its first patient in November 2018, the Wuwei Heavy Ion Center had treated 538 patients with non-small cell lung cancer (NSCLC) as of March 5, 2026. Classified by clinical stage, this cohort includes 165 patients with stage III disease. These 165 cases encompass a variety of NSCLC presentations, including large cell lung carcinoma, as well as cases involving prior treatment such as re-irradiation, post-surgical recurrence, recurrence after radiofrequency ablation, uncontrolled disease after iodine-125 seed implantation, and recurrence after proton therapy. This results in a highly heterogeneous dataset. After excluding these factors to focus solely on patients with primary stage III lung squamous cell carcinoma or adenocarcinoma who had not received prior surgery or radiotherapy, the data becomes clearer, more reliable, and comparable, yielding a total of 134 cases.
Heavy ion therapy was the primary treatment modality for this patient cohort. Generally, depending on the patient's overall condition, specific gene expression, and PD-L1 expression status, concurrent systemic therapy as recommended by the NCCN guidelines was typically advised alongside the heavy ion treatment. This included, for example, a limited course of 1-4 cycles of concurrent or sequential chemotherapy, concurrent or sequential targeted therapy, or concurrent or sequential immunotherapy.
1. Statistics on the Basic Situation of Stage III Non-Small Cell Lung Cancer
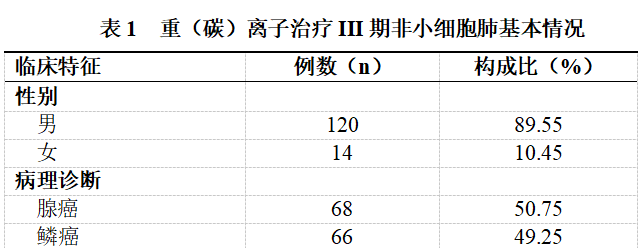
Among the 134 patients with stage III non-small cell lung cancer, there were 120 males and 14 females. The mean age was (63.79 ± 10.83) years, with a maximum age of 85 years and a minimum age of 26 years. Classified by pathological type, there were 68 cases of adenocarcinoma and 66 cases of squamous cell carcinoma. (See Table 1)
2. Survival Analysis of Stage III Non-Small Cell Lung Cancer
(1) Overall Survival Situation
The follow-up period for the 134 patients with stage III non-small cell lung cancer ranged from 0 to 65 months. Survival analysis was performed using the Kaplan-Meier method. The cumulative survival probability at the end of treatment was 1.0000. The cumulative survival probabilities at 3, 6, 12, 24, 36, 48, and 60 months post-treatment were 0.9877, 0.9498, 0.8652, 0.7028, 0.6501, 0.6068, and 0.6068, respectively. (See Table 2, Figure 1)
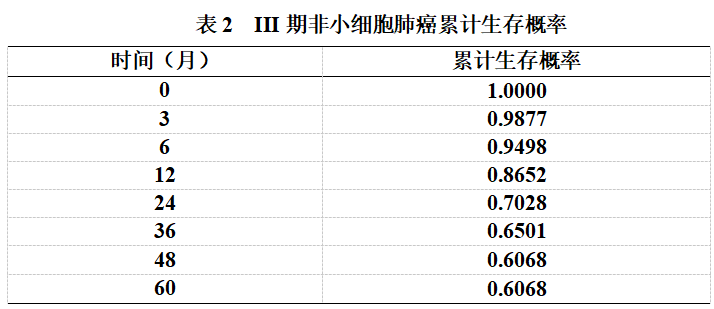
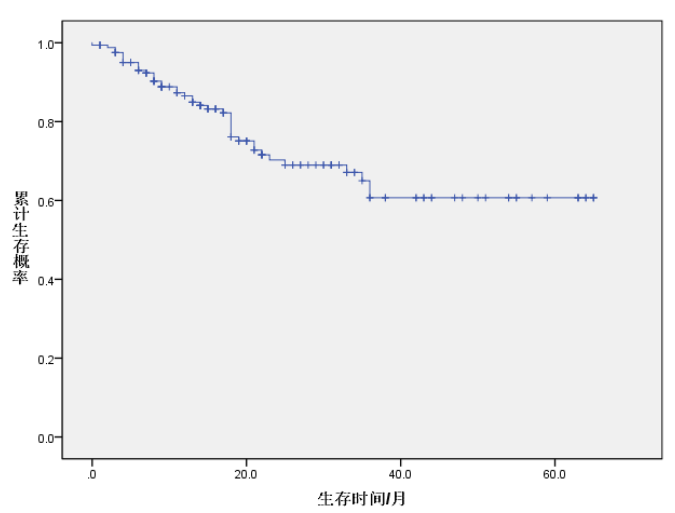
Figure 1. Survival curve from the end of treatment to 60 months post-treatment
(2) Survival status of stage III non-small cell lung cancer with different pathological types (adenocarcinoma, squamous cell carcinoma)
For the 66 patients with stage III non-small cell lung squamous cell carcinoma, the follow-up period ranged from 0 to 65 months. Survival analysis using the Kaplan-Meier method showed a mean survival time of (44.65 ± 3.35) months. The cumulative survival probability at the end of treatment was 1.0000. The cumulative survival probabilities at 3, 6, 12, 24, 36, 48, and 60 months post-treatment were 0.9881, 0.9387, 0.8584, 0.6805, 0.6044, 0.5613, and 0.5613, respectively.
For the 68 patients with stage III non-small cell lung adenocarcinoma, the follow-up period also ranged from 0 to 65 months. Survival analysis using the Kaplan-Meier method revealed a mean survival time of (50.37 ± 3.84) months. The cumulative survival probability at the end of treatment was 1.0000. The cumulative survival probabilities at 3, 6, 12, 24, 36, 48, and 60 months post-treatment were 0.9630, 0.9441, 0.9035, 0.7804, 0.6843, 0.6843, and 0.6843, respectively. (See Table 3, Figure 2)
A Log-rank test was performed, and the survival times of patients with lung squamous cell carcinoma and lung adenocarcinoma were not statistically different (χ²=1.20, P=0.273).
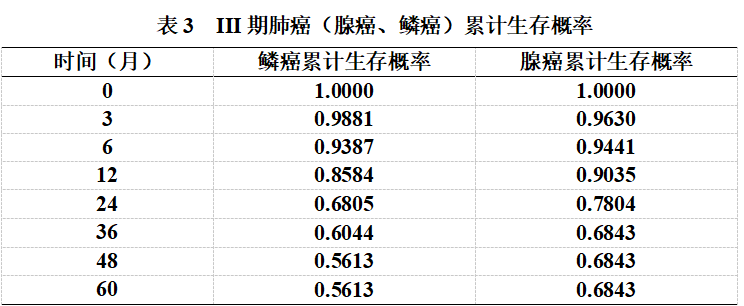
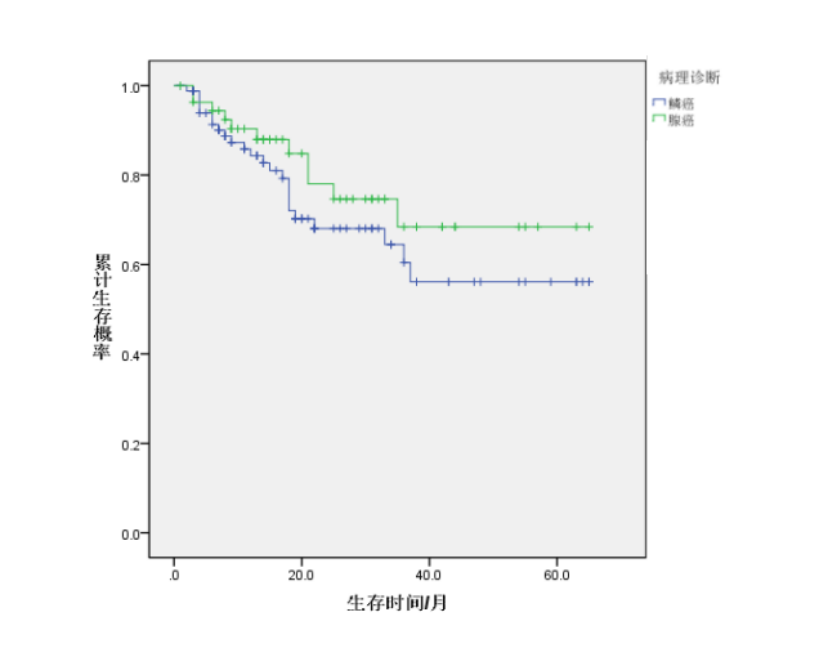
Figure 2. Survival curves from the end of treatment to 60 months post-treatment for patients with squamous cell carcinoma and adenocarcinoma
3. Analysis of Efficacy and Safety
Treatment outcomes for the 134 patients with stage III non-small cell lung cancer at the Wuwei Heavy Ion Center, who received heavy ion-based therapy, show a 2-year overall survival rate of 70.3%, a 4-year overall survival rate of 60.7%, and a 5-year overall survival rate also reaching 60.68%. The median survival time has not yet been reached, as the majority of patients remain alive. These results are significantly superior to those of leading international clinical trials.
For example, in the PACIFIC trial, a landmark study of immunotherapy maintenance following concurrent chemoradiotherapy for stage III NSCLC, the 2-year overall survival rate was 66.3%, the 4-year rate was 49.7%, and the 5-year rate dropped to 42.9%, with a median survival of 47.5 months. In the representative photon-based concurrent chemoradiotherapy study, RTOG 0617, patients receiving 60Gy radiotherapy with chemotherapy had a 2-year overall survival rate of 57.6%, a 4-year survival rate of approximately 30%-40%, and a 5-year survival rate further declining to 20%-30%, with a median survival of only 28.7 months. Meanwhile, a Japanese study on heavy ion (carbon) therapy for stage II-III NSCLC reported a 2-year overall survival rate of 58.7%, a 5-year survival rate of approximately 40%-50%, and a median survival of 29.3 months.
In conclusion, the heavy ion (carbon)-based comprehensive treatment regimen at the Wuwei Heavy Ion Center has achieved excellent efficacy in stage III non-small cell lung cancer, with a 5-year survival rate exceeding 60%. This outcome not only significantly surpasses conventional photon chemoradiotherapy but also outperforms data from similar international heavy ion studies, while offering a better safety profile, higher patient quality of life, and prolonged survival. These findings confirm the substantial advantages and long-term survival benefits of heavy ion (carbon) therapy for stage III non-small cell lung cancer.
4. Answers to Patients' Most Concerned Questions
Q1: Is heavy ion (carbon) therapy truly effective for locally advanced lung cancer?
A: Clinical data from 165 patients with stage III lung cancer treated at the Wuwei Heavy Ion Center show a 5-year survival rate of 60.68%, significantly surpassing the 30%~50% achieved with conventional chemoradiotherapy. The 1-year and 2-year overall survival rates reached 86.5% and 70.2%, respectively.
Q2: I've heard that conventional radiotherapy can cause radiation pneumonitis. Does heavy ion (carbon) therapy also cause radiation pneumonitis?
A: Due to the precise Bragg peak characteristics of heavy ion (carbon) therapy, the radiation dose delivered to normal lung tissue is extremely low. Consequently, the incidence of radiation pneumonitis is significantly lower than with traditional radiotherapy. Data from the Wuwei Heavy Ion Center indicates that the rate of grade 3 or higher radiation pneumonitis is very low; most cases are mild and manageable grade 1-2 reactions, with grade 3 or higher being extremely rare. In many instances, pneumonitis observed in patients is more likely related to immunotherapy or chemotherapy. For experienced heavy ion (carbon) therapy physicians, radiation pneumonitis from this modality is highly manageable.
Q3: Which has better outcomes with this therapy, adenocarcinoma or squamous cell carcinoma?
A: Data from the Wuwei Heavy Ion Center shows a 60-month survival rate of 68.4% for adenocarcinoma and 56.1% for squamous cell carcinoma, with no statistically significant difference (P > 0.05). Heavy ion (carbon) therapy challenges the traditional notion that squamous cell carcinoma carries a poorer prognosis, demonstrating comparable efficacy for both pathological types.
Q4: Is the treatment process painful? How long is the hospital stay?
A: Heavy ion (carbon) therapy is non-invasive and painless. Each treatment session typically lasts only 10-20 minutes, allowing patients to maintain their normal daily lives throughout the treatment course. The Wuwei Center has performed the highest number globally of treatments completed within a single day per fraction. The length of hospital stay varies depending on the individual patient's condition but is generally completed within 30 days.
Q5: Is the treatment expensive? Are there discounts or research programs available?
A: The Wuwei Heavy Ion Center continuously conducts multiple clinical research studies. Eligible patients meeting the study criteria may be eligible for heavy ion (carbon) therapy with zero out-of-pocket costs.
5. Conclusion: Heavy (carbon) ions bring new hope for locally advanced non-small cell lung cancer
Exceptional Efficacy: The 5-year survival rate of 60.68% for stage III lung cancer not only far surpasses that of traditional photon radiotherapy but also exceeds the data from comparable international heavy ion (carbon) studies.
High Safety Profile: Grade 3 or higher toxicities and side effects are extremely rare and manageable, with radiation pneumonitis being mild and controllable.
Technological Leadership: Home to China's first independently developed heavy ion system, a national technological marvel, and backed by 8 years of clinical experience, the center leads the advancement of heavy ion (carbon) technology in China.
Advanced Treatment Model: Employing a comprehensive patient management approach centered on heavy ion (carbon) therapy, the team possesses proficient English language skills (listening, speaking, reading, writing, and translation), ensuring practices are synchronized with and maintain a leading edge internationally.
As the "Bragg peak" of heavy ion (carbon) therapy precisely targets tumors, and as "comprehensive treatment" and "whole-life-cycle management" are integrated throughout the care journey, the Wuwei Heavy Ion Center redefines the meaning of "locally advanced" cancer. It transforms a once-limited prognosis into a new gateway for patients, opening doors to long-term survival and a high quality of life.
6. Wuwei Heavy Ion Center
The Wuwei Heavy Ion Center serves as the clinical application base for China's first independently developed heavy ion (carbon) therapy system. It is technically supported by the Institute of Modern Physics, Chinese Academy of Sciences, and clinically operated by Gansu Wuwei Cancer Hospital. Since commencing clinical treatments in November 2018, the center has treated over 2,800 patients, with its therapeutic applications expanding to more than 50 tumor types, including lung cancer, pancreatic cancer, liver cancer, glioma, nasopharyngeal carcinoma, rectal cancer, and various sarcomas.
As the world's only medical institution simultaneously operating two heavy ion (carbon) therapy systems, Wuwei Cancer Hospital has achieved eight "global firsts" in technological innovation. These pioneering achievements include: the development of precision treatment techniques under ventilator control, bladder cancer therapy with precise bladder volume management, and single-session treatment utilizing surgically placed spacer pads between organs and tumors; the development of China's first 360° rotating and lifting ion therapy chair; and the world's first completion of multiple heavy ion (carbon) therapy cases for cardiac tumors.
Leveraging over 30 research platforms, including the CAS Key Laboratory of Heavy Ion Radiation Biology and Medicine and the University of Sydney's Biomedical Data Analysis and Visualization Laboratory, the center has initiated more than 30 clinical studies focusing on major disease types. To date, the center has published nine professional monographs, including the "Standard Operating Procedures for Carbon Ion Radiotherapy," establishing a comprehensive knowledge framework encompassing physics technology, clinical diagnosis and treatment, and health management.
Contributor: Chen Xuelian (Tumor Registration and Follow-up Office)
Reviewer: Zhang Yanshan (Director of Heavy Ion Center)
Time: March 13, 2026
