Treatment Options for Pancreatic Cancer: Surgery or Heavy (Carbon) Ions? A Comprehensive Overview!
Treatment Options for Pancreatic Cancer: Surgery or Heavy (Carbon) Ions? A Comprehensive Overview!
Author: Zhang Yanshan
Since the first heavy (carbon) ion treatment was performed at the Wuwei Heavy Ion Center in 2018, over eight years have passed. During this period, lung cancer has been the most common disease treated at our center, followed by pancreatic cancer as the second most common. We have accumulated extensive treatment experience, particularly for pancreatic cancer, given the limited effective treatment options available. We have gained significant experience managing various advanced-stage cases and those with complex conditions. To better inform the public about the advantages and limitations of heavy ion therapy, we have prepared this article.
Pancreatic cancer is the fifth leading cause of cancer death and is one of the most lethal malignancies. Historically, surgical resection has been the only curative treatment. However, only a small fraction of patients (10%-20%) are eligible for surgical resection at diagnosis due to the absence of locally advanced or metastatic disease. Even after curative-intent resection, the disease frequently recurs, and the 5-year survival rate remains below 20%. For patients with unresectable disease, chemotherapy and chemoradiotherapy are the standard treatment options. However, due to the inherent resistance of pancreatic cancer to these modalities, local control rates are very low. In recent years, along with the development of novel anticancer drugs, more advanced radiotherapy techniques have also been introduced. Nevertheless, these treatments still fail to provide satisfactory outcomes, and the median survival time remains approximately 10 months in most cases.
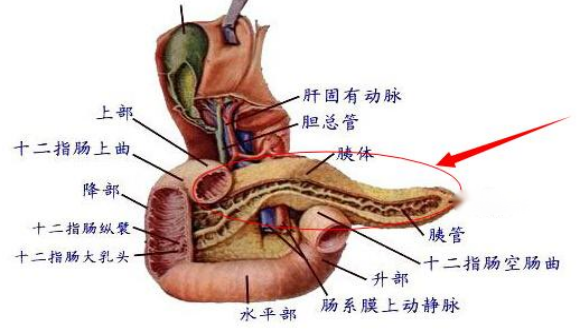
1. Characteristics of pancreatic cancer
Anatomically complex, surrounded by the duodenum, stomach, and colon, deeply located, often asymptomatic in early stages, and easily missed by conventional imaging examinations. Pancreatic cancer tissue is dense, has poor blood supply, and is markedly hypoxic. Additionally, pancreatic cancer exhibits neurotropism and readily invades surrounding nerves. Furthermore, due to the density of the tumor, the tissue becomes ischemic and hypoxic, making it difficult for chemotherapeutic drugs to penetrate. Compared to the oxygen concentration in adjacent normal pancreatic tissue, all pancreatic tumors demonstrate significant hypoxia. The resistance of cancer cells in a hypoxic environment to chemotherapy and radiotherapy is 2–3 times greater than that of cells under normoxic conditions.
2. Advantages and disadvantages of pancreatic cancer surgery
The advantage of surgery is the ability to remove the primary tumor along with the closely associated duodenum and surrounding lymph nodes in one procedure. This is the strength of surgical treatment for pancreatic cancer, especially for cancers of the pancreatic head. If complete resection is achievable, surgery should be the priority. For patients with early-stage disease without distant metastasis, surgical resection offers the potential for long-term survival and even a cure. The postoperative specimen allows for definitive TNM staging, assessment of differentiation grade, and evaluation of lymph node status. This pathological staging is more accurate than radiological staging and provides the most reliable basis for decisions regarding subsequent adjuvant chemotherapy or radiotherapy.
The disadvantages of surgery lie in its significant trauma and high risk. The risk is substantial due to the complex anatomy, with major blood vessels surrounding the pancreas (such as the superior mesenteric artery and portal vein) making surgical dissection difficult. There is a high risk of intraoperative bleeding and potential damage to critical vessels. Surgery can also easily promote tumor dissemination and metastasis. Furthermore, anastomotic leaks, such as enteric fistulas and pancreatic fistulas, are common and represent the most challenging issues in pancreatic surgery. Due to the soft texture of the pancreas and the corrosive nature of pancreatic juice, healing of the pancreaticoenteric anastomosis is difficult. Should a pancreatic fistula occur, it can lead to abdominal infection and hemorrhage, and is a primary cause of postoperative mortality. The rate of achieving an R0 resection (microscopically negative margins) is also limited, as tumors frequently encase the superior mesenteric artery or portal vein; even with vascular resection and reconstruction, ensuring no cancer cells remain at the margins is difficult. Moreover, at the time of diagnosis, many patients, despite imaging showing disease apparently confined to the pancreas, already harbor micrometastases in their bloodstream. Following resection of the primary tumor, these micrometastases can rapidly develop into distant metastases (e.g., to the liver) and peritoneal dissemination. Consequently, postoperative recurrence rates are high. Due to the highly aggressive biological behavior of pancreatic cancer, a significant proportion of patients experience local recurrence, peritoneal seeding, or liver metastases within months to 1-2 years after even curative-intent resection. Therefore, surgery typically needs to be combined with postoperative adjuvant chemotherapy to extend survival.
Surgery is only suitable for a select group of patients with early-stage disease and good performance status. Pancreaticoduodenectomy places high demands on a patient's cardiopulmonary function and nutritional status; elderly patients or those with severe comorbidities may not tolerate the procedure. Forcing surgery in patients with locally advanced or metastatic disease is not only unhelpful but may also diminish quality of life due to surgical trauma.
In short, the advantage of surgery lies in its potential for a cure, while its disadvantages are high risk and a propensity for recurrence. The key to successful surgery lies not only in resecting the tumor but also in precise preoperative assessment (selecting the right patients for resection), meticulous intraoperative technique (reducing the risk of pancreatic fistula), and comprehensive postoperative multimodality therapy (chemotherapy/targeted therapy/immunotherapy).
3. Advantages of heavy ion radiation therapy
In heavy ion radiotherapy, heavy (carbon) ion therapy has a nearly equivalent lethal effect on hypoxic cancer cells and normoxic cancer cells. Heavy ions are more effective at killing hypoxic cells. Furthermore, although pancreatic cancer stem cells have been shown to be resistant to conventional chemotherapy and radiotherapy, heavy ion beams demonstrate a stronger killing effect on these cells. This may be because heavy ion radiotherapy causes more double-strand breaks in DNA compared to X-rays. Heavy ion beams are also safer and more effective than conventional radiotherapy methods. A complex nerve plexus surrounds the main arteries of the pancreas, an area where local recurrence occurs in most pancreatic cancer cases after treatment. Heavy ion beams possess a unique depth-dose profile, which offers a potential advantage by enabling the safe delivery of a biologically effective dose to the tumor while sparing adjacent organs at risk, such as the stomach and duodenum (which are close to the pancreas and sensitive to radiation), thereby protecting these critical structures.
4. Irradiation range of heavy (carbon) ion therapy for pancreatic cancer
The advantages of ion therapy are also reflected in its larger irradiation field, which can cover the tumor itself, the nerve plexus, and regional lymph nodes. Regional lymph nodes include the celiac lymph nodes, superior mesenteric lymph nodes, hepatoduodenal lymph nodes, splenic artery lymph nodes, and peripancreatic lymph nodes, among others.
5. Heavy (carbon) ion therapy for locally advanced pancreatic cancer
For locally advanced unresectable pancreatic cancer, the determination that the tumor cannot be resected is based on CT and MRI findings showing tumor encasement of the celiac trunk or superior mesenteric artery (corresponding to stage III according to TNM classification). Heavy (carbon) ion therapy is administered once daily, with a total of 12 fractions delivered over 3 weeks. Toxicities are generally mild. The main acute toxicities observed are anorexia and cholangitis. To further improve efficacy, concurrent heavy ion therapy and chemotherapy is a favorable approach, with manageable side effects. The 2-year local control rate is 58%, and the 2-year overall survival rate is 54%. This significantly surpasses the 10%-25% 2-year survival rate reported in the literature for other treatment modalities used for locally advanced pancreatic cancer. Therefore, concurrent heavy ion therapy and chemotherapy has become the optimal treatment strategy for locally advanced pancreatic cancer.
Typical Case: Patient Hu, male, 61 years old, diagnosed with malignant neoplasm of the pancreas, adenocarcinoma, cT4N2M1 stage IV. The patient reported that when he was hospitalized at a major center, he joined a surgical treatment group for pancreatic cancer patients. Most patients in that group underwent chemotherapy, surgery, further chemotherapy, plus immunotherapy and targeted therapy; almost none lived longer or had a better quality of life than him. In the four years following his initial heavy (carbon) ion therapy, he was able to play soccer and frequently engaged in strenuous exercise. No one would believe he was a pancreatic cancer patient. After recurrence, his performance status declined, and he could no longer play soccer. He received his first course of heavy (carbon) ion therapy in November 2021, with a total dose of 55.2 Gy (RBE) delivered in 12 fractions to the primary pancreatic lesion, and a total dose of 60 Gy (RBE) delivered in 10 fractions to the metastatic lesion in liver segment S5. He received a second course of heavy (carbon) ion therapy in February 2025, with a dose of 60 Gy (RBE) in 15 fractions. He received a third course of carbon ion therapy in October 2025, with a dose of 60 Gy (RBE) in 15 fractions, concurrently with chemotherapy. As of March 17, 2026, his performance status is poor. This case involved three courses of heavy (carbon) ion therapy for pancreatic cancer.
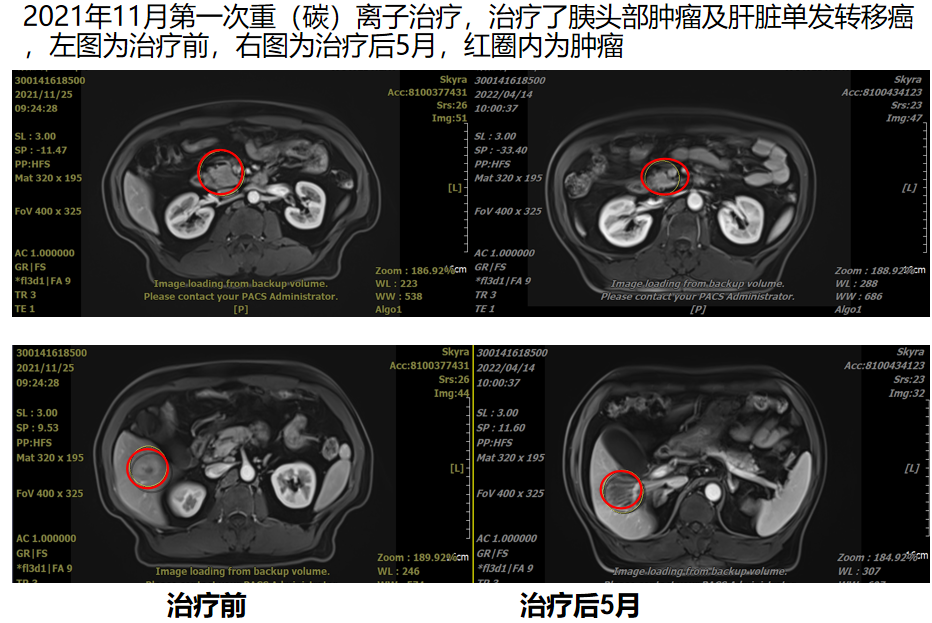
The figure shows: November 2021, first heavy (carbon) ion therapy, treating the pancreatic head tumor and a solitary liver metastasis.
The left image shows the condition before treatment, and the right image shows the condition 5 months after treatment. The red circles indicate the tumors.
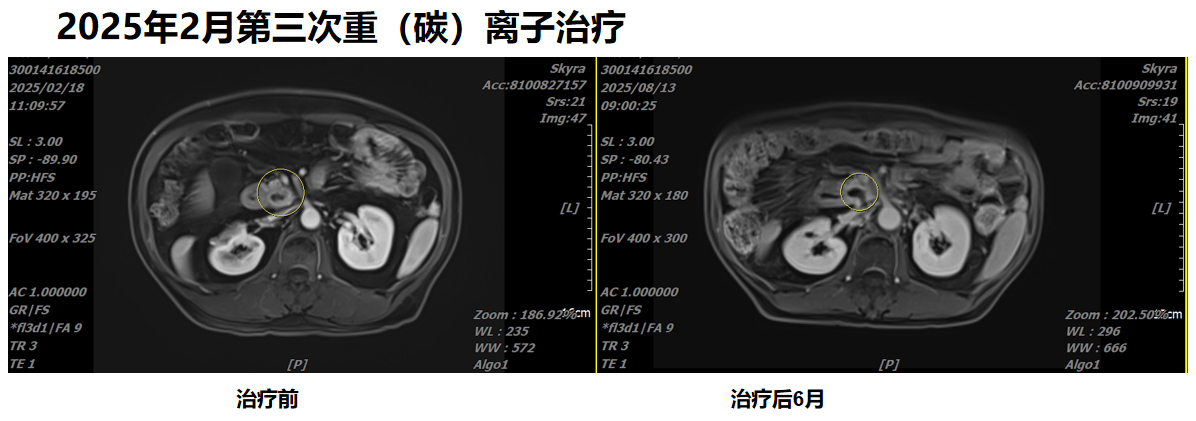
The figure shows a comparison before the third heavy (carbon) ion therapy in February 2025 and six months after treatment.
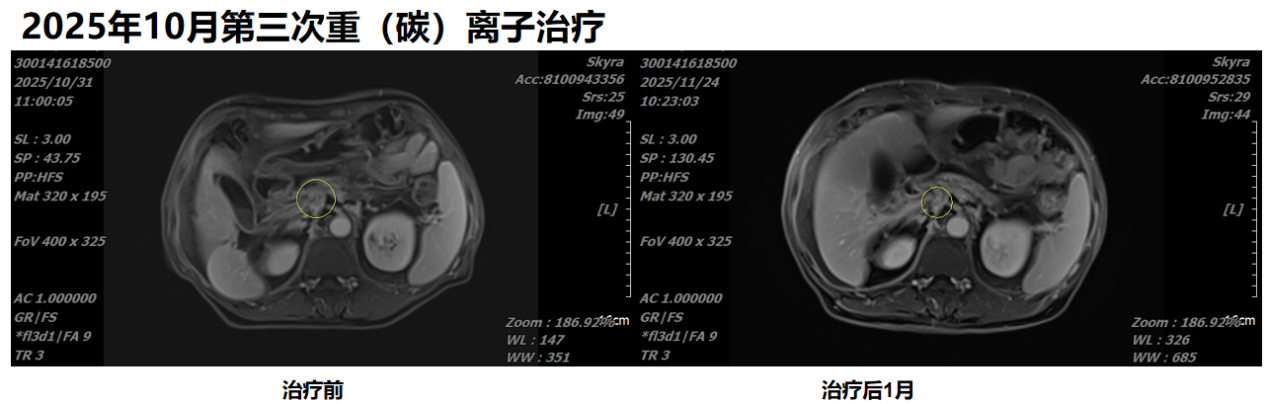
The figure shows a comparison before the third heavy (carbon) ion therapy in October 2025 and one month after treatment.
Case provided by: Director Zhang Yihe
Department: Radiation Oncology Center Ⅰ(International Medical Department Ⅰ)
6. Comparison of heavy ion therapy with proton therapy and photon therapy (including CyberKnife, TOMO, etc.)
The killing effect of heavy ions on pancreatic cancer cells is 3–5 times greater than that of proton or photon therapy. Proton therapy and photon therapy generally target only the tumor itself. However, as we know, pancreatic cancer tends to invade nerves and metastasize to lymph nodes. The treatment volume for heavy (carbon) ion therapy includes the tumor, most or even the entire pancreas, thereby encompassing the nerves and regional lymph nodes that pancreatic cancer commonly invades. Another key point is that pancreatic cancer itself does not respond well to proton therapy and photon therapy; its inherent characteristics make it less sensitive to these modalities.
7. Choose surgery or heavy ions?
Before the advent of heavy ion therapy, surgery was the only modality offering hope for long-term survival. However, only a small fraction of patients with early-stage disease (10%-20%) are eligible for surgical resection at diagnosis due to the absence of locally advanced or metastatic spread. Furthermore, even after curative-intent resection, the disease frequently recurs, and the 5-year survival rate remains below 20%. The majority of pancreatic cancer patients are diagnosed at a stage where surgical treatment is no longer feasible. Compounding this issue, pancreatic cancer exhibits poor sensitivity to chemotherapy, resulting in low response rates. Consequently, so-called "conversion therapy" (administering chemotherapy first with the aim of subsequent surgery) often delays effective treatment and may worsen the prognosis. Therefore, timely selection of heavy ion therapy combined with chemotherapy represents an effective strategy. However, a significant challenge exists for cancers in the pancreatic head. Since the pancreatic head is largely surrounded by the duodenum, despite the high sensitivity of pancreatic cancer cells to heavy ions, there is a concern for radiation-induced injury to the bowel. This often necessitates limiting the heavy ion dose to the portion of the tumor adjacent to the intestine, which is a primary reason for suboptimal outcomes in some pancreatic cancer cases treated with heavy (carbon) ion therapy. One often sees reports of excellent results following heavy (carbon) ion therapy for pancreatic cancer; in these cases, the tumor's location is typically at some distance from the bowel, allowing physicians to deliver a higher heavy ion dose, leading to excellent efficacy. In contrast, for pancreatic head cancer, the deliverable heavy ion dose is often constrained. Consequently, the curative potential of heavy (carbon) ion therapy for pancreatic head cancer is somewhat limited. To address this issue, it is advisable to employ techniques commonly used at the Wuwei Heavy Ion Center, such as the SPACER technique and the HFOV technique. Otherwise, for pancreatic head cancer, surgery is the preferred approach whenever feasible. For patients in whom surgery is not possible, these techniques can be considered. If surgery is truly not an option, heavy ion therapy becomes the best alternative, and when combined with concurrent chemotherapy, it represents the current optimal treatment regimen. For tumors located in the pancreatic body and tail, the difficulty and risks of surgical resection are significantly lower. Moreover, a higher heavy ion dose can potentially be delivered. For these patients, the efficacy of heavy ion therapy is generally very good.
8. The necessity of postoperative heavy (carbon) ion therapy
One of the benefits of heavy ion therapy is its mild toxicity and minimal side effects. It is also highly sensitive to lymphatic metastases. Therefore, for patients found to have positive lymph nodes after pancreatic surgery, timely administration of postoperative heavy ion radiotherapy to the pancreatic tumor bed and the retroperitoneal lymphatic drainage area can significantly prevent or delay metastasis and recurrence, generally without compromising the patient's quality of life.
9. Iodine seed implantation therapy
In my over thirty years of medical practice, I have personally performed and overseen iodine seed implantation for the treatment of various solid tumors. During my graduate studies, I also followed my supervisor's guidance to perform iodine seed implantation for cancer patients, giving me a profound understanding of this modality. It could be said that iodine-125 seed implantation, in a certain way, reflects the individualized and sometimes non-standardized nature of oncological treatment in China. Iodine-125 seed implantation is a form of brachytherapy. The procedure involves implanting radioactive iodine-125 seeds into the tumor via a minimally invasive (percutaneous puncture) method or during surgery. As the iodine-125 seeds decay, they emit low-dose gamma rays, providing continuous, uninterrupted irradiation to the tumor tissue. This can kill tumor cells at different stages of the cell cycle as well as hypoxic cells surrounding the tumor. The gamma rays emitted by iodine seeds are a type of photon. A major issue with this technique is that the implantation requires puncture with numerous large needles, which carries a risk of needle-track seeding, and the treatment volume is quite limited. According to the latest international NCCN guidelines for the treatment of common solid tumors, only a few specific diseases, such as prostate cancer, are considered suitable for iodine-125 seed implantation. Clinically, many patients are concerned about needle-track dissemination, and they are often anxious even about diagnostic biopsies. Accepting iodine seed implantation for pancreatic cancer is particularly problematic, as it often leads to poor outcomes and rapid peritoneal dissemination. Both physicians and patients need to fully consider this issue and must be very well-informed. If a physician requires a specific radiation dose to the area targeted by the iodine seed implantation puncture, this dose can be safely delivered using our standard external beam radiotherapy techniques.
10. The necessity and importance of chemotherapy
Pancreatic cancer has a high propensity for liver metastasis and peritoneal dissemination. To prevent metastasis and improve local efficacy, chemotherapy for pancreatic cancer is necessary and remains critically important.
11. The necessity and importance of biopsy
In clinical practice, if a pathological diagnosis for pancreatic cancer is not available, a clinical diagnosis can potentially be established based on imaging findings combined with serum biomarker tests. In such cases, timely chemotherapy and heavy (carbon) ion therapy can be initiated. However, it should be emphasized that, whenever possible, obtaining a pathological diagnosis for a tumor is highly recommended. This is crucial for the overall assessment of the disease and for guiding prognosis. Furthermore, at experienced institutions like our center, painless endoscopic ultrasound-guided fine-needle aspiration biopsy of the pancreas can be readily performed. This procedure is simple, efficient, and highly recommended.
12. Genetic testing and immunological testing
Overall, the probability of pancreatic cancer harboring clinically actionable driver gene mutations or exhibiting high PD-L1 expression or MSI-H status is relatively low. However, approximately 3%-5% of patients can still benefit from corresponding targeted therapies or immunotherapies. For pancreatic cancer, this is still a valuable opportunity, as we know that, in comparison, the response rate to various chemotherapy regimens for pancreatic cancer is only around 20%-30%. Despite the significant toxicities associated with chemotherapy, it remains a standard treatment that almost all patients receive.
13. Can pancreatic cancer with liver metastasis be treated with heavy (carbon) ion therapy?
For pancreatic cancer patients presenting with a solitary or limited number of liver metastases (oligometastasis), treatment with heavy (carbon) ion therapy directed at both the primary pancreatic tumor and the liver metastases, combined with concurrent chemotherapy, can be beneficial. Several of my own patients with pancreatic cancer and liver metastases have achieved complete tumor regression and long-term survival following this approach of heavy ion therapy with concurrent chemotherapy. However, if the liver involvement is characterized by diffuse, widespread metastases, the potential benefit is limited.
14. Can pancreatic cancer with peritoneal metastasis be treated with heavy (carbon) ion therapy?
For patients with peritoneal metastasis, the cancer cells have already disseminated into the peritoneal cavity. In this situation, heavy ion therapy cannot provide benefit and is not a suitable treatment option. More relevant therapeutic approaches include TIL (Tumor-Infiltrating Lymphocyte) cell immunotherapy combined with chemotherapy and intraperitoneal perfusion. Overall, the efficacy of treatment for peritoneal metastasis is poor, and survival outcomes are limited. At our center, the TIL immunotherapy process involves collecting mononuclear cells from ascites fluid, isolating the immune T cells capable of recognizing the tumor from within these cells, expanding them in vitro, and then reinfusing them back into the patient.
15. Heavy ion therapy has a significant effect on patients with pancreatic cancer complicated by extensive retroperitoneal lymph node metastasis and pain
The significant back pain caused by pancreatic cancer is often due to retroperitoneal metastatic lymph nodes compressing the lumbar sympathetic trunk, combined with the neurotropic nature of pancreatic cancer cells, which readily infiltrate and grow along the nerves. Heavy ion therapy is particularly effective in this scenario, as it can rapidly alleviate pain and significantly improve the patient's quality of life, fully leveraging the advantages of heavy ion therapy.
16. Main Toxic and Side Effects of Heavy (Carbon) Ion Therapy for Pancreatic Cancer
Overall, the toxicities and side effects associated with heavy (carbon) ion therapy for pancreatic cancer are very mild, and patients typically do not perceive significant adverse effects from the radiation itself. However, when combined with chemotherapy, the side effects from the chemotherapy remain quite pronounced. Specifically, for pancreatic body or tail cancers treated with heavy (carbon) ion therapy, patients generally experience virtually no side effects from the radiation. In contrast, for pancreatic head cancer, the tumor often compresses the pancreatic duct and bile duct. Edema induced by the radiotherapy can further obstruct these ducts, leading to impaired drainage of bile and pancreatic juice. This can result in symptoms such as anorexia, cholangitis, and even pancreatitis in some patients.
Summary
In summary, heavy ion therapy is a highly effective, low-toxicity local treatment modality. Its efficacy is approximately 2-5 times greater than that of proton therapy, CyberKnife, or TOMO therapy, and it is fundamentally different from proton therapy. Most patients experience no side effects during treatment, allowing it to be considered a non-invasive, highly effective alternative to surgery. It is highly sensitive for various pancreatic tumors. However, it is crucial to correctly recognize that heavy ion therapy is only a local treatment. One must avoid blindly exaggerating its efficacy. Only through scientifically sound patient selection and appropriate application can patients derive the maximum benefit.
Reviewer: Zhang Yanshan (Vice President of Gansu Wuwei Cancer Hospital)
Data Providers: Li Pengqing, Chen Xuelian (Cancer Registration and Follow-up Office)
Date: March 18, 2026
