Ten Years of Companionship, Looking Forward to a Longer Future
Ten Years of Companionship, Looking Forward to a Longer Future
– A Case Study on the Treatment of Small Cell Lung Cancer
How many decades are there in a person's life? At Wuwei Cancer Hospital, there is a patient with advanced lung cancer who has undergone intermittent treatment for ten years. This case gives us a deeper understanding and insight into the treatment of cancer patients. We also hope that our friends battling cancer can find more courage and hope in their long journey of living with the disease, adhere to standardized treatments, and live a life of quality and hope.
Case Study
In October 2013, our hospital admitted Mr. Li, a patient from Guangdong with small cell lung cancer. He came to our hospital's Department of Thoracic Surgery due to symptoms of "irritating dry cough and shortness of breath." A chest CT scan revealed a space-occupying lesion in the anterior mediastinal thymic area (42mm × 35mm × 32mm), multiple nodular lesions in the right lung, and destructive changes in the T12 vertebral body. An MRI of the head and lumbar spine showed no significant tumor lesions.
In early November 2013, he underwent an "exploratory right thoracotomy" under general anesthesia. The pathological examination indicated small cell lung cancer, with cancerous tissue infiltration found in the (third) rib and the submitted diaphragmatic tissue. After surgery, the patient underwent three cycles of chemotherapy, and a follow-up examination showed a reduction in the lesions. Subsequently, the patient did not receive further treatment due to financial reasons. On November 1, 2016, a follow-up chest CT scan indicated disease progression (PD). Concurrent radiotherapy and chemotherapy were recommended, but the patient and family refused radiotherapy and instead underwent six intermittent cycles of chemotherapy.
From June to October 2018, due to disease progression, the patient received another four cycles of chemotherapy. A follow-up chest CT scan showed stable disease, but the patient discontinued chemotherapy due to financial constraints. From November 9, 2018, to September 2019, the patient participated in a clinical trial conducted by our department and was treated with "Compound Taxus Capsule 0.6g orally three times a day." Regular follow-ups showed stable disease. However, a chest CT scan in August 2019 indicated disease progression, and the trial was concluded. Due to financial reasons, the patient received intermittent chemotherapy at our hospital from July 2020 to January 2023. During this period, due to poor compliance, the patient did not undergo regular chemotherapy.
In December 2021, the patient developed brain metastases and received two-dimensional conformal radiotherapy for the mediastinal tumor and brain metastases from December 16 to December 28, 2021. In January 2023, the disease progressed again due to irregular chemotherapy. Unwilling to continue chemotherapy, the patient started oral targeted therapy with "Afatinib Maleate Tablets (self-provided) 40mg once daily," which has continued to the present. On March 3, 2023, a follow-up non-contrast CT scan of the chest, abdomen, and pelvis, along with a non-contrast MRI of the head, showed stable disease (SD).
This patient has now safely passed ten years, living and working as usual. His family is healthy, and he is surrounded by children and grandchildren.
Every time we contact him, we are reminded that life is long, and it is enough to be happy and alive. Our department will continue to follow up with this patient. We hope every patient can live a quality life filled with happiness.
Treatment-related imaging data:
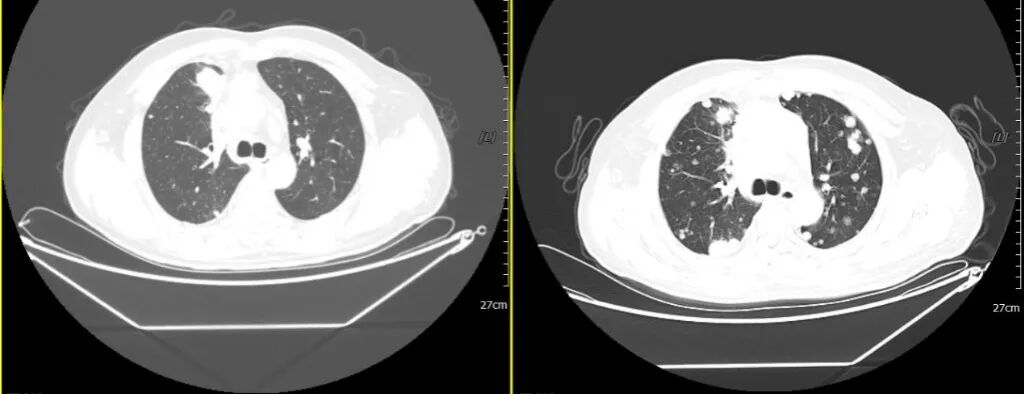
2020-07-03 2021-02-20
Comparison with the CT scan from July 3, 2020: Multiple abnormal nodules in both lungs have significantly increased in number and size compared to before (previously approximately 3.2×2.1 cm, now approximately 3.9×3.0 cm). Enlarged lymph nodes in the anterior mediastinal space, mediastinum, and both hila have partially increased in size compared to before (previously approximately 5.2×3.4 cm, now approximately 6.7×4.0 cm), with sternal involvement (new finding). Involvement of the adjacent superior vena cava and left brachiocephalic vein has worsened compared to before.
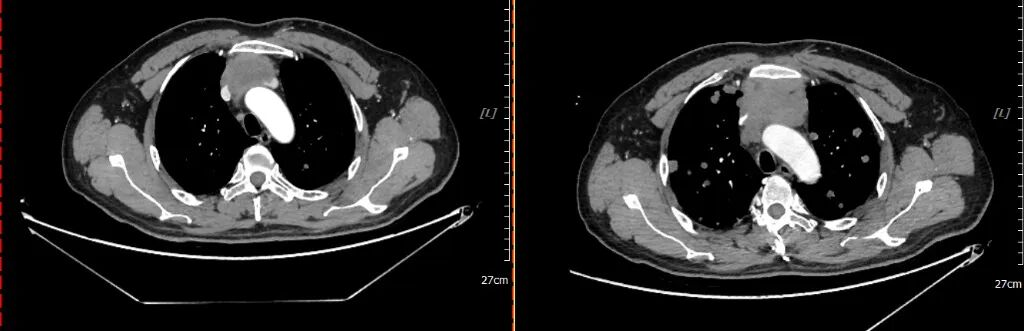
2021-02-20 2021-04-30
Re-examination for efficacy evaluation after 2 cycles of chemotherapy: PR (Partial Response)
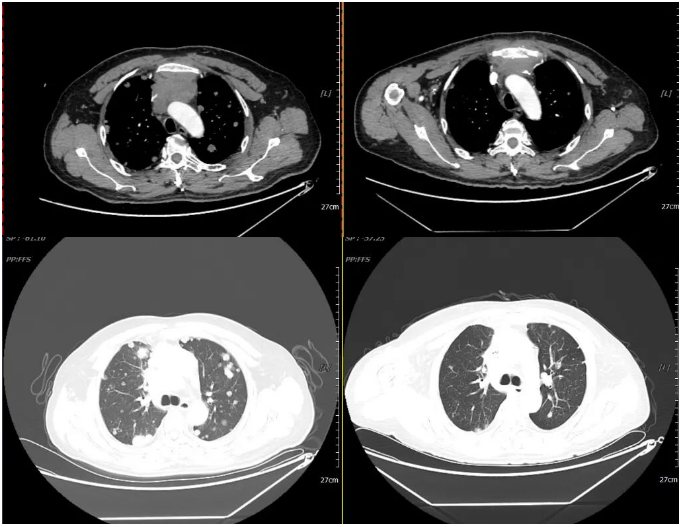
2021-04-30 2021-07-06
Re-examination for efficacy evaluation after 4 cycles of chemotherapy: SD (Stable Disease) (nodules in both lungs and pleura have decreased in number and size; lymph nodes in the anterior mediastinal space, mediastinum, and both hila (2.5×3.0 cm) have partially reduced in size compared to before).
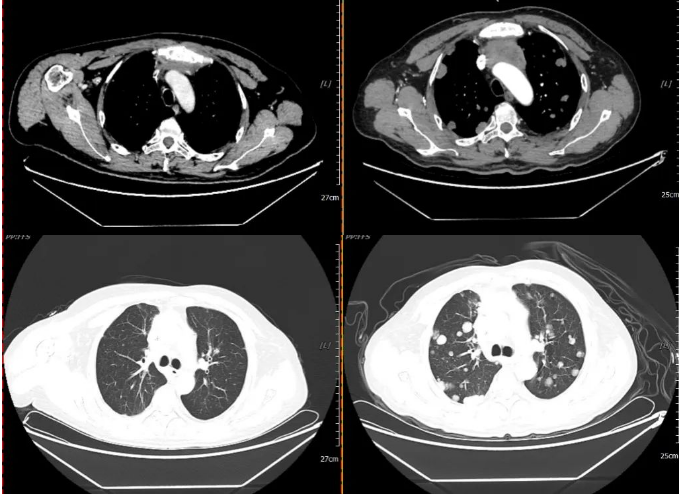
2021-07-06 2021-12-11
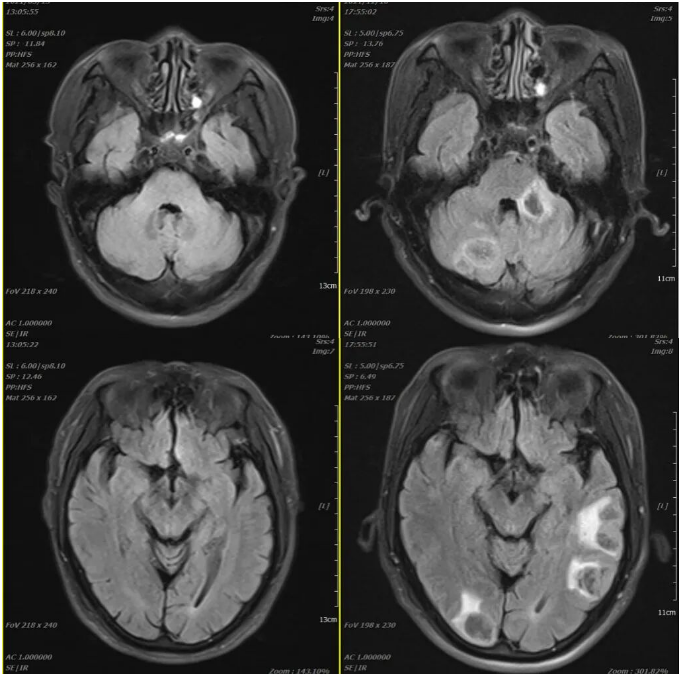
2021-03-05 2021-12-10
Efficacy evaluation on December 11, 2021: PD (Progressive Disease) (nodules in both lungs and pleura have significantly increased in number and size; metastatic lymph nodes in the anterior mediastinal space, mediastinum, and both hila (now approximately 4.7×3.5 cm) have increased in number and size compared to before, with involvement and narrowing of the superior vena cava and left brachiocephalic vein; new metastases in both cerebellar hemispheres and cerebral hemispheres (approximately 2.1×1.7 cm)).
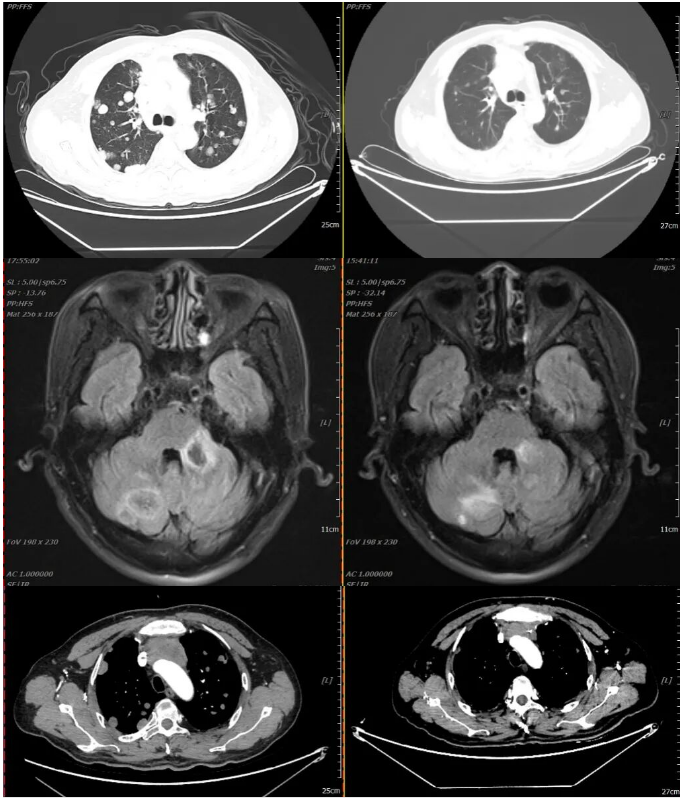
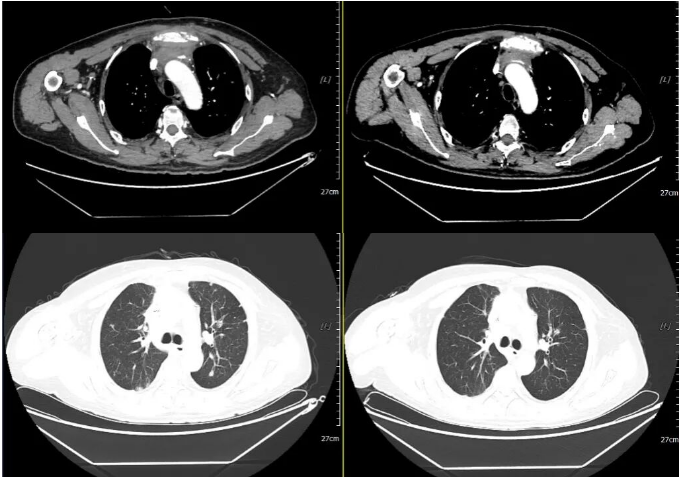
2021-12-11 2022-03-23
Oral targeted therapy with "Afatinib Maleate Tablets (self-provided) 40mg orally once daily" was initiated on January 30, 2023.
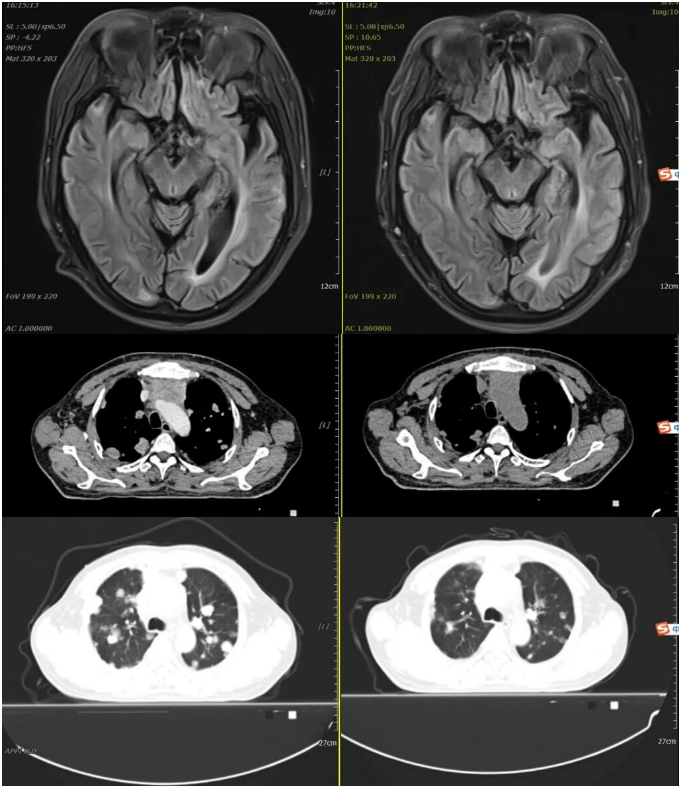
2023-01-30 2023-03-03
Re-examination at our hospital on March 3, 2023, with non-contrast CT of the chest, abdomen, and pelvis, and non-contrast MRI of the head: Efficacy evaluation: SD (Stable Disease).
Introduction to the Chemotherapy Department
The Chemotherapy Department of Wuwei Medical Science Academy Cancer Hospital was established in February 2009. It currently has 46 beds. Under the leadership of Director Wang Huijuan, Chief Physician, the department now has 7 doctors, including 1 Chief Physician, 1 Associate Chief Physician, 2 senior attending physicians, and 3 resident physicians. The department has long-term appointments with Vice President Zhao Da, a chief expert in medical oncology from the First Hospital of Lanzhou University, as an external consultant for regular ward rounds and academic exchanges. The department admits over 1,200 patients for chemotherapy annually, making it the largest and most standardized medical oncology department in the Hexi region. In 2013, it was designated as one of the first "Standardized Cancer Pain Management Demonstration Wards" in Gansu Province. In 2017, the team sent by the department won the first prize in the "Gansu Provincial Standardized Cancer Pain Management Skills Competition". In 2017, it became a member of the "Gansu Provincial Lung Cancer Prevention and Treatment Alliance". In 2018, it became a member of the "Lanzhou University Comprehensive Cancer Treatment Specialist Alliance", and its team won the third prize in the "2019 Gansu Provincial Lung Cancer MDT Competition". In 2021, it was recognized as a "Wuwei City Key Discipline". In July 2022, it became a "Sub-center of the Gansu Provincial Chest Tumor Clinical Medicine Research Center". The department currently provides the following technologies: diagnosis of various solid tumors and lymphomas, neoadjuvant chemotherapy, postoperative adjuvant chemotherapy, palliative chemotherapy, and local drug perfusion chemotherapy; endocrine therapy; tumor molecular targeted therapy, tumor immune checkpoint inhibitor therapy, and cellular immunotherapies such as CIK and NK. The department leads and participates in numerous drug clinical trials, with well-completed trials bringing benefits to patients.

Contact Us
Contact Number: 0935-6988068, 0935-6988063
Medical Address: Department of Chemotherapy, Gansu Wuwei Cancer Hospital (2nd Floor, Rehabilitation Building), Xuanwu Street, Liangzhou District, Wuwei, Gansu Province, China
