Radionuclide Therapy – Explore Radionuclide Therapy Solutions Today
Radionuclide Therapy – Explore Radionuclide Therapy Solutions Today
 Doctor, I have been diagnosed with terminal cancer and am facing a heart-wrenching decision between continuing treatment or stopping treatment to return home. Could you offer me some guidance?
Doctor, I have been diagnosed with terminal cancer and am facing a heart-wrenching decision between continuing treatment or stopping treatment to return home. Could you offer me some guidance?
 Many cancer patients believe that a terminal diagnosis means the countdown has begun, with no hope for a cure—but that’s not always the case. In fact, with standardized and appropriate treatment, some types of cancer can still achieve meaningful remission, even at an advanced stage.
Many cancer patients believe that a terminal diagnosis means the countdown has begun, with no hope for a cure—but that’s not always the case. In fact, with standardized and appropriate treatment, some types of cancer can still achieve meaningful remission, even at an advanced stage.
 But the problem is, I have pancreatic cancer—the so-called "the king of cancers" If I continue treatment, will I end up with both my life and money lost in the end?
But the problem is, I have pancreatic cancer—the so-called "the king of cancers" If I continue treatment, will I end up with both my life and money lost in the end?
 For many advanced cancer patients who have exhausted conventional treatments like radiotherapy, chemotherapy, targeted therapy, and immunotherapy, radionuclide therapy in nuclear medicine may emerge as a new beacon of hope.
For many advanced cancer patients who have exhausted conventional treatments like radiotherapy, chemotherapy, targeted therapy, and immunotherapy, radionuclide therapy in nuclear medicine may emerge as a new beacon of hope.
Today, let’s explore together how to choose the most suitable radionuclide therapy for your specific case.

Lutetium-177-TBM therapy for bone metastases
IndicationsIndications
Bone metastases in the terminal stage of various cancers including lung cancer, breast cancer, prostate cancer, esophageal cancer, ovarian cancer, pancreatic cancer, liver cancer, renal cancer, and gallbladder cancer.
Background
Bone metastasis is a common complication in most advanced cancers. Patients often experience bone pain, myelosuppression, and pathological fractures, which significantly impact quality of life and overall survival.
Treatment Options
Lutetium-177-TBM (¹⁷⁷Lu-TBM) is a precision-targeted radionuclide therapy for bone metastases. Compared with conventional treatments, it offers the following advantages:
● Simplified Procedure: The treatment process is straightforward with high patient compliance.
● Precise Targeting: Selectively acts on metastatic bone lesions while minimizing impact on surrounding healthy tissues.
● Minimal Side Effects: Causes no significant myelosuppression and has low adverse reaction rates.
● Proven Efficacy: Demonstrates remarkable pain relief, particularly effective for patients with widespread bone metastases.
Security
Lutetium-177-TBM (¹⁷⁷Lu-TBM) therapy demonstrates reliable safety:
● Targeted Efficacy: Specifically acts on bone metastases without damaging healthy organs and tissues.
● Low Dosage: Therapeutic efficacy can be achieved with just 30 mCi of the radiopharmaceutical.
● Low Toxicity: Minimal risk of myelosuppression with no significant impact on hepatic or renal function.
In summary, Lutetium-177-TBM (¹⁷⁷Lu-TBM) represents a safe and effective therapeutic option for bone metastases, with its clinical benefits outweighing potential risks.
Classic Case Report:

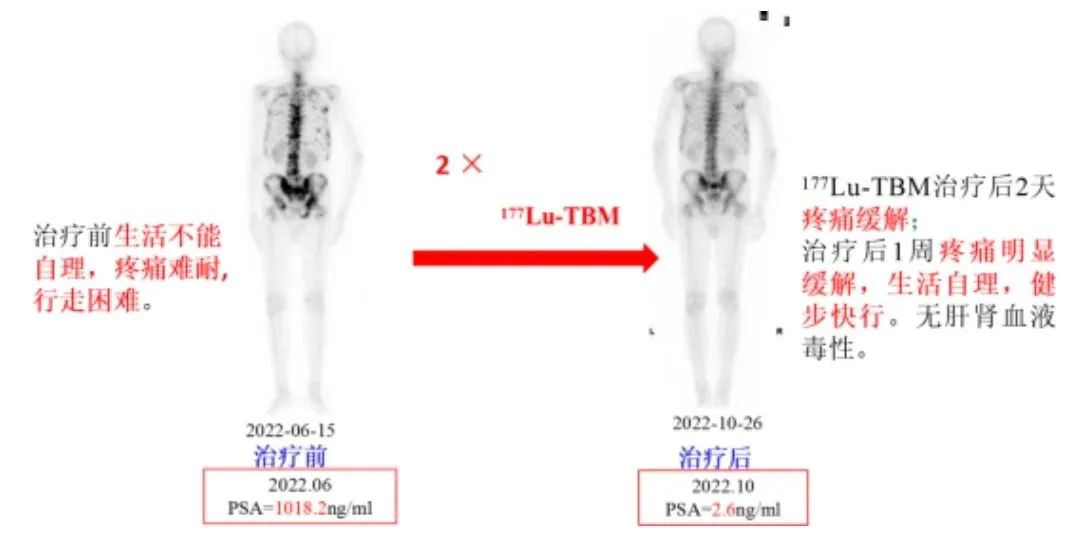
Lutetium-177-PSMA therapy for prostate cancer
Indications
Advanced prostate cancer, specifically metastatic castration-resistant prostate cancer (mCRPC), which refers to lethal prostate cancer that continues to progress and metastasize despite androgen deprivation therapy.
Background
Prostate cancer is a highly prevalent malignant tumor. While endocrine therapy can effectively control the disease, patients often develop resistance over time. Additionally, prostate cancer frequently metastasizes to bones, significantly increasing treatment difficulty. Moreover, elderly patients generally have poor tolerance for conventional radiotherapy and chemotherapy.
Mechanism
Targeting the prostate cancer-specific biomarker PSMA, this approach achieves precision treatment by conjugating the radionuclide lutetium-177 with PSMA. This therapeutic method offers new hope for advanced prostate cancer patients, particularly those resistant to endocrine therapy.
Security
Both domestic and international studies have demonstrated that lutetium-177-PSMA therapy is associated with minimal hematologic toxicity, exhibiting a favorable overall safety profile with only a minority of patients experiencing adverse effects.
Classic Case Report:
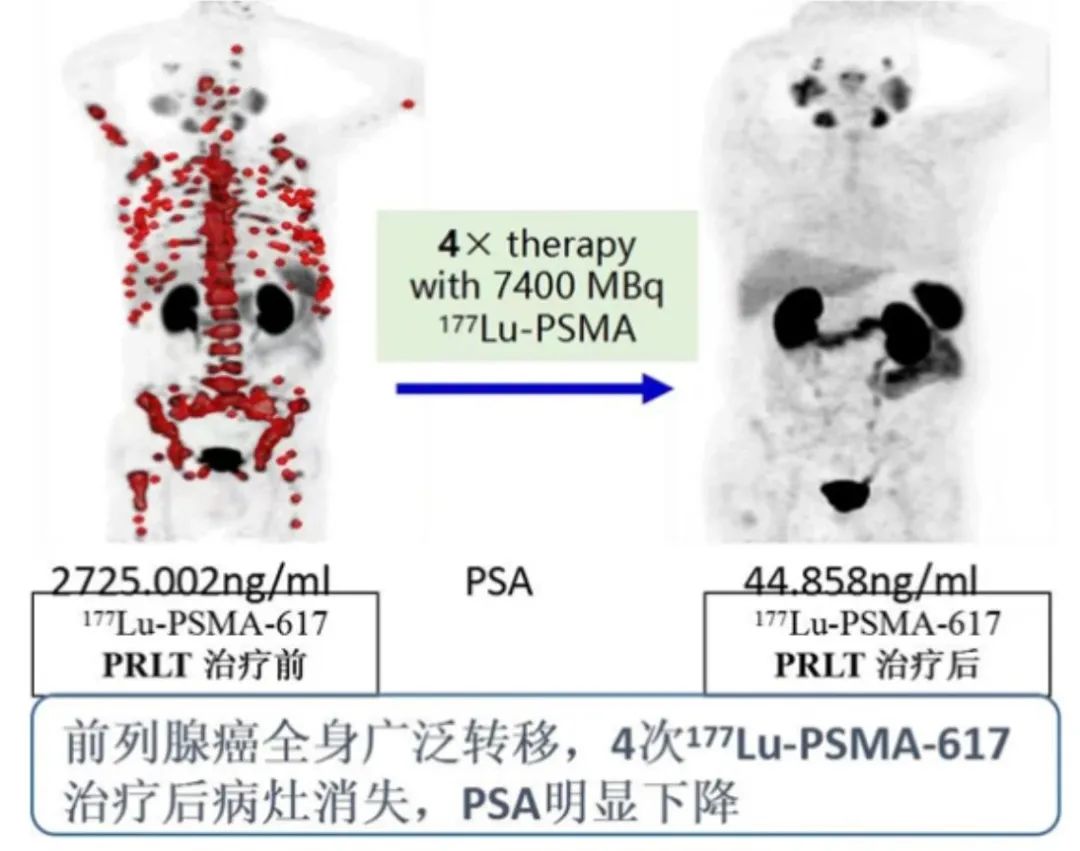
Lutetium Lu 177 Dotatate for the Treatment of Neuroendocrine Tumors
Indications
Indicated for patients with SSTR2-positive metastatic or unresectable neuroendocrine tumors (NETs), including gastrointestinal, pancreatic, hepatic, and rectal NETs, as well as bronchopulmonary carcinoids, pheochromocytomas, paragangliomas, neuroblastomas, and medullary thyroid carcinomas.
Background
Neuroendocrine tumors (NETs) are relatively rare neoplasms originating from neuroendocrine cells distributed throughout the body. Characterized by neuroendocrine markers and the ability to produce bioactive amines or peptide hormones, their incidence has been steadily increasing. These tumors may arise in any organ or tissue, including endocrine organs (e.g., thymus, pituitary, adrenal glands) and non-endocrine organs (e.g., lungs, gastrointestinal tract, pancreas). Gastro-entero-pancreatic neuroendocrine tumors (GEP-NETs) represent the most common subtype, accounting for approximately two-thirds of all NET cases.
Therapeutic applications
Lutetium Lu 177 Dotatate is primarily indicated for G1 and G2 grade neuroendocrine tumors (NETs) with positive PET/CT findings, and may also be considered for G3 patients (typically requiring combination therapy). It is generally employed as a second-line or later treatment option for advanced refractory NETs, applicable when conventional therapies—including biologic therapies (e.g., SSAs), molecular targeted agents, or chemotherapy—have failed.
Security
Both domestic and international studies consistently demonstrate that only a minimal proportion of patients experience hematologic toxicity or other adverse reactions during lutetium-177-DOTATATE therapy.
Classic Case Report:
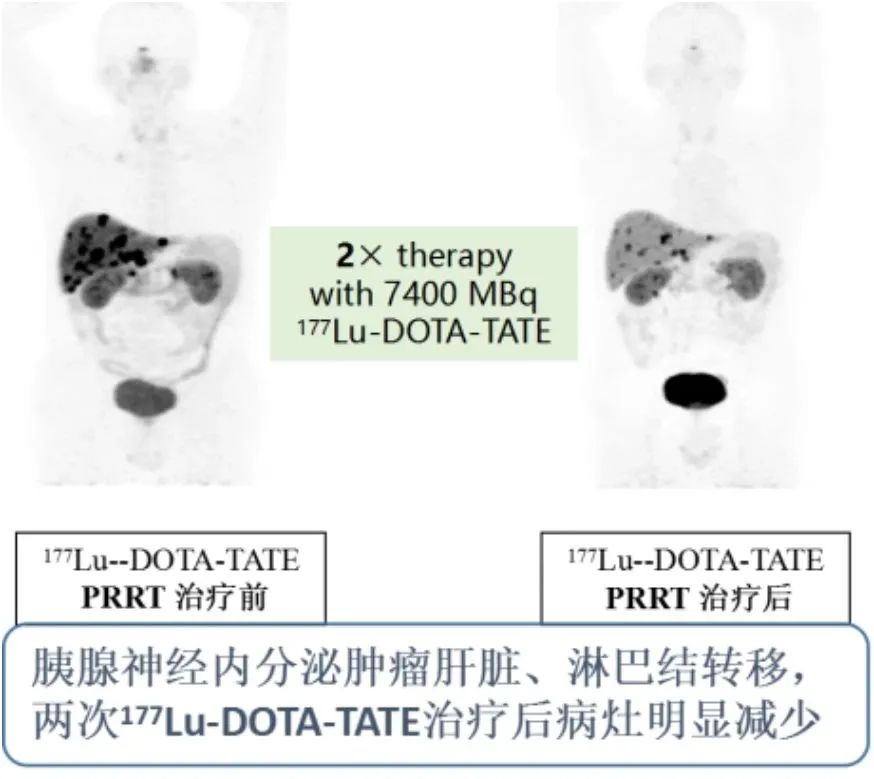
Lutetium Lu 177 FAP-2286 for the Treatment of Advanced Solid Tumors
Indications
Indicated for various epithelial-origin tumors, including breast cancer, lung cancer, pancreatic cancer, cholangiocarcinoma, ovarian cancer, rectal cancer, meningioma, and melanoma.
Background
FAP (Fibroblast Activation Protein) is closely associated with tumor desmoplastic reactions. Normal fibroblasts exhibit little to no FAP expression, whereas tumor-associated fibroblasts (TAFs) demonstrate significant FAP overexpression—particularly in breast, colorectal, pancreatic, and lung cancers with marked desmoplasia, where fibroblasts constitute up to 90% of the tumor stroma. FAP-2286 exhibits enhanced FAP affinity, higher tumor uptake, and prolonged intratumoral retention compared to earlier agents.
Therapeutic applications
Lutetium-177-FAP-2286 has demonstrated significant therapeutic efficacy with minimal side effects in treating various advanced cancers, including pancreatic cancer, breast cancer, ovarian cancer, and rectal cancer.
Classic Case Report:

 After reviewing all this information, do you have any further questions?
After reviewing all this information, do you have any further questions?
 I understand that I have pancreatic cancer with bone metastases, and the bone pain is particularly severe. The lutetium-177 TBM (Tetraazacyclododecane Tetraacetic Acid-Bone Metastasis) therapy in nuclear medicine seems suitable for me. However, besides bone metastases, I also have lymph node and liver metastases. Would I be a candidate for lutetium-177 FAP-2286 therapy?
I understand that I have pancreatic cancer with bone metastases, and the bone pain is particularly severe. The lutetium-177 TBM (Tetraazacyclododecane Tetraacetic Acid-Bone Metastasis) therapy in nuclear medicine seems suitable for me. However, besides bone metastases, I also have lymph node and liver metastases. Would I be a candidate for lutetium-177 FAP-2286 therapy?
 Indeed, our Nuclear Medicine Department consistently adheres to the theranostic concept in personalized patient care. We first utilize radionuclide imaging to precisely evaluate lesion uptake of targeted agents, thereby selecting the most suitable treatment regimen for each individual patient.
Indeed, our Nuclear Medicine Department consistently adheres to the theranostic concept in personalized patient care. We first utilize radionuclide imaging to precisely evaluate lesion uptake of targeted agents, thereby selecting the most suitable treatment regimen for each individual patient.
 Regular chemotherapy is really draining. I barely recover from one hospital visit before needing to go back again, and the side effects are so severe that they leave me extremely weak with drastically reduced quality of life. So how often would this radionuclide therapy be administered?
Regular chemotherapy is really draining. I barely recover from one hospital visit before needing to go back again, and the side effects are so severe that they leave me extremely weak with drastically reduced quality of life. So how often would this radionuclide therapy be administered?
 Radionuclide therapy is typically administered in cycles every 6 to 8 weeks. After completing two cycles, an imaging scan will be performed to evaluate treatment response. Generally, the side effects during and after treatment are mild. However, for advanced cancers, a comprehensive approach combining multiple therapies is often necessary. Even with radionuclide therapy, other treatments like radiation or chemotherapy should not be discontinued. Only through this multimodal strategy can we achieve optimal disease control and the best possible outcomes.
Radionuclide therapy is typically administered in cycles every 6 to 8 weeks. After completing two cycles, an imaging scan will be performed to evaluate treatment response. Generally, the side effects during and after treatment are mild. However, for advanced cancers, a comprehensive approach combining multiple therapies is often necessary. Even with radionuclide therapy, other treatments like radiation or chemotherapy should not be discontinued. Only through this multimodal strategy can we achieve optimal disease control and the best possible outcomes.
 Is this treatment really the so-called "miracle therapy" that works with just one injection? It sounds almost too remarkable to be true.
Is this treatment really the so-called "miracle therapy" that works with just one injection? It sounds almost too remarkable to be true.
 It's not just a simple injection, but the entire procedure is straightforward and usually takes about 2-3 hours. The treatment is typically administered through an IV infusion, or alternatively using an IV pump - the process feels similar to receiving antibiotics intravenously.
It's not just a simple injection, but the entire procedure is straightforward and usually takes about 2-3 hours. The treatment is typically administered through an IV infusion, or alternatively using an IV pump - the process feels similar to receiving antibiotics intravenously.
