Liver Cancer Screening: Which of the Four Key Tests Best Reflect Liver Health?
Liver Cancer Screening: Which of the Four Key Tests Best Reflect Liver Health?
Overview
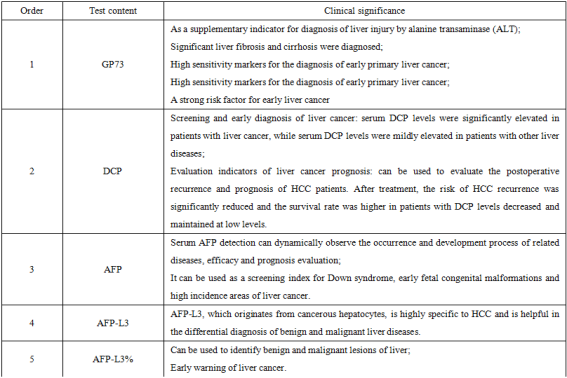
Golgi protein 73 (GP73) is a transmembrane glycoprotein localized on the Golgi apparatus, consisting of 402 amino acids with a relative molecular mass of 73 kDa. In healthy livers, GP73 is expressed at low levels. However, during chronic liver conditions such as inflammation, injury, fibrosis, or cirrhosis, GP73 expression progressively increases with disease severity and decreases upon remission. This dynamic pattern makes GP73 a novel serological marker for evaluating the progression of liver diseases.
Abnormal prothrombin, also known as Protein Induced by Vitamin K Absence or Antagonist-II (PIVKA-II) or Des-γ-carboxyprothrombin (DCP), is a protein closely linked to vitamin K deficiency. Normally, prothrombin (a clotting factor) is synthesized by hepatocytes. However, in patients with hepatocellular carcinoma (HCC), a key feature is systemic vitamin K deficiency. Vitamin K is essential not only for synthesizing coagulation factors II (prothrombin), VII, IX, and X but also acts as a cofactor in the conversion of fibrinogen to fibrin during blood clotting. When vitamin K is deficient, hepatocytes produce large amounts of abnormal prothrombin (DCP/PIVKA-II) due to impaired carboxylation. This results in coagulation dysfunction, clinically manifested as bleeding tendencies. Quantifying DCP/PIVKA-II levels can therefore serve as a biomarker to assess hepatocyte damage and monitor liver disease progression.
Alpha-fetoprotein (AFP) is a fetal-specific serum glycoprotein synthesized by ribosomes on the rough endoplasmic reticulum of human hepatocytes. In hepatocellular carcinoma (HCC), serum AFP levels are markedly elevated. However, elevated AFP is not exclusive to HCC and may also occur during pregnancy or in other malignancies, including embryonal carcinoma, ovarian cancer, testicular cancer, and occasionally gastric, pancreatic, or colorectal cancers. Despite these non-specific elevations, the dynamic monitoring of AFP levels (e.g., persistent rise or values >400 ng/mL) holds significant clinical value in diagnosing HCC, particularly when combined with imaging findings and risk factor assessment (e.g., cirrhosis or chronic hepatitis).
Alpha-fetoprotein (AFP) can be classified into three subtypes based on its binding affinity to Lens culinaris agglutinin (LCA): AFP-L1, AFP-L2, and AFP-L3.
AFP-L1: Primarily observed in healthy individuals or those with benign liver diseases.
AFP-L2: Mainly produced by the yolk sac and commonly elevated during pregnancy.
AFP-L3: Generated predominantly by hepatocellular carcinoma (HCC) cells.
During hepatocellular carcinogenesis, the sugar chains of AFP undergo structural modifications, specifically fucosylation (addition of fucose residues). This fucosylated AFP exhibits higher binding affinity to LCA and is termed the AFP-L3 isoform. AFP-L3 levels correlate strongly with tumor size, differentiation grade, and malignancy severity, demonstrating superior specificity compared to total AFP in diagnosing HCC.
AFP-L3% (also referred to as the AFP-L3 isoform ratio) represents the percentage of AFP-L3 relative to total alpha-fetoprotein (AFP) in serum.
Intended population
(1) Individuals with fatty liver or liver fibrosis;
(2) Chronic liver diseases caused by alcohol, chemical toxins, or drug use;
(3) Genetic metabolic liver diseases, infectious liver diseases, or liver cancer patients;
(4) Viral hepatitis carriers or autoimmune hepatitis patients;
(5) People with long-term alcohol consumption, irregular sleep patterns, or unhealthy lifestyles;
(6) Routine screening for general health check-up populations.
Test indicators
The four primary biomarkers for liver cancer screening commonly include: Golgi Protein 73 (GP73), Des-gamma-carboxy Prothrombin (DCP), Alpha-fetoprotein (AFP), AFP-L3 Isoform (AFP-L3) and its ratio (AFP-L3%).
Sample submission requirements
(1) Both outpatient and inpatient applications are available. Project instructions: four items of liver cancer, or abnormal prothrombin (PIVKA-II) test (single item) can also be issued separately.
(2) Sample requirements: 3ml peripheral blood should be collected using a procoagulant tube (yellow cap tube). Samples that cannot be tested on the same day should be stored at 2-8℃ for 24 hours. Samples should be stored at-20℃ after 24 hours, and samples should not be repeatedly frozen and thawed.
(3) Report is issued on the same day. Outpatients print reports with their own printers at the guidance desk, and inpatients print reports with the hospital LIS system.
Biological chip center
The Biochip Center offers the following testing services: Genetic Testing (e.g., deafness gene screening, newborn early screening) Pathogen Detection (e.g., respiratory pathogenic bacteria, respiratory viruses, SARS-CoV-2 nucleic acid testing), Tumor Gene Testing (e.g., ALK, ROS1, NRAS, HRAS, PIK3CA, MET, RET, HER-2), Pharmacogenomics (e.g., clopidogrel medication guidance, warfarin dosing guidance, statin therapy guidance, proton pump inhibitor optimization, hypertension drug-related gene testing).





A glimpse of the gene biochip room and the biological immune cell therapy room
The Department of Biological Immunotherapy is pioneering state-of-the-art tumor immunotherapies. Currently, we are conducting clinical research on advanced cellular therapies, including: Cytokine-Induced Killer (CIK) Cells,Natural Killer (NK) Cells,Dendritic Cell-CIK (DC-CIK) Therapy, Umbilical Cord Mesenchymal Stem Cell Therapy, additionally, we are planning to initiate clinical trials for Chimeric Antigen Receptor T-Cell (CAR-T) Therapy in the near future.
