Efficacy observation of carbon ion radiotherapy for adenoid cystic carcinoma of head and neck
Efficacy observation and literature review of carbon ion radiotherapy for adenoid cystic carcinoma of head and neck
Li Xiaojun1, Chai Hongyu1*, Zhang Yanshan1, Zhou Junnian2, Hu Tingchao1, Ma Shuping1, Zhang Tiane1, Wang Yan1, Wang Jian1
1Heavy ion radiotherapy department,2Head and neck surgery department, Wuwei Cancer hospital of Gansu province, Wuwei Gansu 733000, China.
[Abstract] Objective To analyze and discuss the advantages of carbon ion radiotherapy in the treatment of head and neck adenoid cystic carcinoma (ACC) through 5 cases report and literature review of the head and neck ACC treated with carbon ion radiotherapy. Materials and methods Retrospectively report the case data, treatment methods and equipment, simulation positioning process, target delineation, follow-up plan, efficacy and adverse reactions of 5 patients with the head and neck adenoid cystic carcinoma who received carbon ion radiotherapy in our center from 2020 to 2021.The prescribed dose to the target area is 64.0-68.0Gy[Relative Biological Effectiveness (RBE)] (RBE=3.0)/16-17Fx. Results All 5 patients had completed carbon ion radiotherapy. The average follow-up period was 15.8 months (13.0-20.0 months). At 12 months of follow-up, 1 patient achieved complete response (CR), and 4 patients achieved partial response(partial response, PR), 1 patient was evaluated as progressive disease (PD) due to pulmonary metastasis after treatment for 6 months. The 12-month overall survival (OS) was 100%, the local control (LC) was 100%, and the progression free survival (PFS) was 80%. Grade 2 acute oral mucosal reaction occurred in 1 case (case 4), and 1 late adverse reaction to radiotherapy was observed, manifesting as grade 1 dry mouth, and no toxic reaction of grade 3 or above occurred in all patients. Conclusion Carbon ion radiotherapy has good survival rate and local control rate for unresectable, postoperative residual and recurrent ACC of the head and neck, and the toxic and side effects are tolerable.
[Key words] head and neck, adenoid cystic carcinoma, carbon ion therapy, efficacy, adverse reactions
Adenoid cystic carcinoma (ACC), characterized histopathologically by epithelial nests containing hyaline material forming "cylindrical" patterns, is alternatively termed cylindroma or adenocarcinoma of cylindroma type. First described by Billroth in 1859, ACC represents a relatively rare malignant tumor with an annual incidence of 3.0-4.4 cases per million population.
The head and neck region constitutes the most frequent site of ACC occurrence, accounting for approximately 1% of all head and neck malignancies [1]. Predilection sites include minor salivary glands, sublingual glands, parotid glands, and submandibular glands, with ACC representing 24% of salivary gland malignancies [2]. Less common locations encompass the nasopharynx, oral floor, bronchi, and parapharyngeal spaces [3]. The disease demonstrates no significant gender predominance, with peak incidence occurring between 40-60 years of age [4].
The insidious nature and aggressive biological behavior of head and neck ACC often lead to multi-organ involvement due to proximity to oral, nasal, ocular, and skull base structures. The tumor exhibits remarkable infiltrative growth patterns, with microscopic tumor extension frequently observed beyond grossly normal tissue margins, sometimes demonstrating discontinuous "skip" metastases. Although characterized by relatively indolent progression, ACC demonstrates marked propensity for perineural invasion and hematogenous dissemination [5]. These clinicopathological features frequently result in advanced disease at diagnosis, diagnostic challenges, low cure rates, and unsatisfactory long-term outcomes.
Surgical resection remains the primary therapeutic modality for head and neck ACC. However, complete excision with adequate margins is often unattainable, with reported 10-year local control rates of merely 60% following surgery, decreasing below 30% in margin-positive cases [6-7]. The frequent involvement of multiple functional anatomical compartments and extensive subclinical spread render many tumors unresectable or amenable only to palliative debulking procedures with high recurrence rates.
Consequently, radical radiotherapy has emerged as a crucial component in the multidisciplinary management of ACC. As a radioresistant neoplasm, ACC demonstrates a dose-response relationship for local control, yet dose escalation with conventional photon radiotherapy remains constrained by adjacent organ tolerance limits [8]. Reported 5-year overall survival rates following photon radiotherapy stand at only 37.3%, underscoring the radioresistant nature of ACC [9]. These limitations necessitate more effective treatment modalities capable of improving local control and survival while preserving critical normal structures.
Our institution's carbon ion therapy system commenced clinical operation in March 2020. This report presents preliminary clinical outcomes of five head and neck ACC patients treated with carbon ion radiotherapy, including analyses of local control rates, overall survival, progression-free survival, and treatment-related toxicity profiles.
1. Materials and Methods
1.1 Patient Characteristics
The study cohort comprised five consecutive patients with histologically confirmed head and neck ACC treated with carbon ion therapy at our institution between March 2020 and June 2021. All patients underwent comprehensive pretreatment evaluation including contrast-enhanced MRI/CT and PET-CT imaging. The mean patient age was 57.2 years (range: 35-77 years).
Primary tumor distribution:
- Maxillary sinus: 2 cases
- Nasal cavity: 1 case
- Parotid gland: 1 case
- Sinonasal cavity: 1 case
Disease staging according to AJCC 8th edition:
- Stage II: 1 case
- Stage III: 1 case
- Stage IVA: 2 cases
- Stage IVB: 1 case
Treatment background:
- Case 1: Postoperative recurrence
- Cases 2-3: Postoperative residual disease
- Cases 4-5: Medically inoperable disease
All patients exhibited good performance status with ECOG scores of 0-1.
Table 1 General patient information
| Case No. | Age | Gender | Primary Location | Invasion Sites | Histopathology | TNM Stage | Follow-up (months) |
| 1 | 58 | male | Parotid gland | Carotid sheath | ACC | pT4aN0M0 IVA stage | 20 |
| 2 | 77 | female | Maxillary sinus | No | ACC | pT2N0M0 II stage | 19 |
| 3 | 52 | male | Maxillary sinus | Right hard palate | ACC | pT3N0M0 III stage | 17 |
| 4 | 35 | male | Paranasal sinuses | Maxillary sinus wall, orbit | ACC | cT4aN2cM0 IVB stage | 14 |
| 5 | 64 | male | Nasal cavity | Bilateral pterygopalatine fossa | ACC | cT4bN0M0 IVA stage | 13 |
1.2 Treatment methods and equipment
All patients completed head and neck enhanced nuclear magnetic, PET-CT, head and neck enhanced CT and other related examinations before treatment, and individualized carbon ion treatment plan was formulated after multidisciplinary consultation in our center.CT localization equipment is Philips Brilliance CT Big Bore large aperture CT, Wuwei carbon ion medical gas pedal HIMM was developed by Lanzhou Institute of Modern Physics of the Chinese Academy of Sciences. Configuration of radiation treatment planning system ci-Plan was independently developed by the Institute of Modern Physics, which is suitable for the design of high-energy carbon ion beam radiation treatment plan.
1.2.1 Simulation positioning method
Adopt supine position, open mouth with mouth plug, press the tongue under the mouth plug, reduce the area of the tongue irradiated and reduce the area of irradiated oral mucosa irradiated by opening the mouth. The headrest was selected individually according to the patient's specific situation, and the head and neck were adjusted to the angle according to the site of the lesion for thermoplastic film fixation. After the mold was made, a localization CT scan was performed, the scanning range was from the top of the skull to the level of the tracheal bifurcation, the scanning requirement was 1mm thin-layer CT, and the localization MRI scan was performed every other day, the requirement was a thin-layer 1mm MRI scan with the mold and the mouth gag. At the end of the scanning, the localization CT and MRI will be fused to refer to the principle of head and neck carbon ion target area outlining for target area outlining, MRI is routinely used to identify the tumor, and PET-CT is referred to when necessary. total target volume (GTV) efficacy determination of the maximum diameter determination based on MRI enhancement.
1.2.2 Methods of target area outlining
Primary tumor volume (GTV) included tumor lesions shown by enhanced MRI and/or PET/CT examination, including retropharyngeal lymph nodes.GTVnd for neck metastatic lymph nodes, outlining for neck metastatic lymph nodes shown by enhanced MRI and/or PET/CT examination. The planned target volume, PTVboost, was defined as a 5-mm expansion in all directions on the basis of GTV and GTVnd. Clinical target volume (CTV) included GTV+3-5 mm and the extent of subclinical foci based on the surgical and histopathological reports and MRI or enhanced CT images, and determined according to the biological behavior of the tumor. adenoid cystic carcinoma is prone to invade along the nerves, so it should include the sites of the tumors nerves and their pathways up to the skull base or intracranial. Prophylactic irradiation of the lymph node drainage area is not routinely performed, and in patients with pre-existing cervical lymph node metastases, prophylactic irradiation of the lymph node drainage area in that region is required. The planned target volume PTV was defined as a CTV based on a 3-mm flare in all directions, taking into account organ motion and posing errors. Segmental dosing was used to calculate the dose distribution of carbon ion passive beam uniform scanning irradiation using ci-Plan, with the first course of planned PTV-Boost: 8-12 Gy(RBE)/2-3 Fx, and the second course of PTV: 54 Gy(RBE)/14 Fx. The prescribed dose to the tumor for the five patients ranged from 64.0 to 68 Gy(RBE)/16 to 17 Fx. Carbon Ion doses are expressed as Gy(RBE), defined as the physical dose multiplied by the carbon ion RBE, all at a relative biological effectiveness (RBE) of 3.0. Carbon ion endangered organ limits refer to the dose limits of each endangered organ for ion therapy throughout the body [10], brainstem Dmax < 45 Gy (RBE), D1 ≤ 38.5 Gy (RBE); crystalline D1 < 6 Gy (RBE); optic pathway D20 < 30 Gy (RBE); spinal cord Dmax < 30 Gy (RBE), D1 ≤ 31.5 Gy (RBE); parotid gland Dmean < 21 Gy (RBE) (bilateral), Dmean < 18 Gy (RBE) (unilateral).
1.3 Follow-up program
The efficacy assessment was performed 3 months after the completion of carbon ion radiotherapy, and the examination items included MRI, CT, and laboratory tests of the relevant sites; CT or MRI was reviewed every 3 months within 18 months after treatment, followed up every 6 months from 18 months to 3 years, and PET-CT was performed once a year for 4-5 years after treatment. Overall survival and progression-free survival were calculated from day 1 of carbon ion radiation therapy. Based on clinical signs and symptoms, test results, acute and late stage during the 5-year follow-up period were evaluated with reference to the Common terminology criteria for adverse events version 5.0 (CTCAE v5.0 ) and EORTC/RTOG radiation injury grading criteria. Adverse events, according to the Rating Criteria for Effectiveness in Solid Tumors (RECIST)
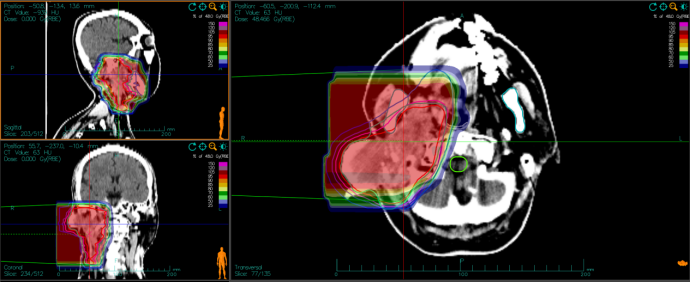
Fig.2 The image of the dose distribution of carbon ion radiotherapy of Case 1.
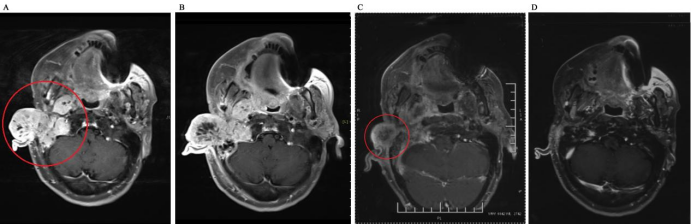
Figure 2 CT images before and after CIRT A. Before treatment; B. SD after treatment; C. PR after 1 month after treatment D. CR after 5 months after treatment

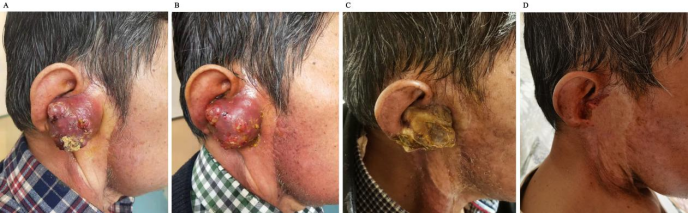
Fig.3 Images of tumor size change before and after carbon ion radiotherapy A. before treatment B. 10 fraction after treatment C. PR after 1 month after treatment D. CR after 5 months after treatment
All observed acute and late radiotherapy adverse reactions are listed in Table 2.No grade 3 or higher radiotherapy-related adverse reactions occurred in all patients. Most of the acute reactions occurred during and within 2 weeks after treatment and manifested as radiation dermatitis, nasal oral mucositis. one case (case 4) experienced a grade 2 oral mucosal reaction, which manifested as moderate pain with ulceration requiring fluid intake. Late adverse reactions to radiotherapy were observed in one case of dry mouth, which appeared 3 months after the end of radiotherapy, and was characterized by dry mouth, loss of taste, and viscous saliva with decreased secretion.
Table 2 Acute and late adverse reactions to radiotherapy
| 级别 | 放疗急性不良反应(例) | 放疗晚期不良反应(例) | ||||||
| 放射性皮炎 | 鼻腔黏膜炎 | 口腔黏膜炎 | 出血 | 口干 | 咽部粘膜炎 | 骨坏死 | 吞咽困难 | |
| 1级 | 3 | 1 | 1 | 0 | 1 | 0 | 0 | 0 |
| 2级 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 |
| 3级 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
3 Discussion.
ACC in the head and neck region is typically characterized by an unpredictable and slow course, complex anatomical relationships, and susceptibility to local recurrence [11]. Surgical treatment requires extensive local resection, and since adequate margins cannot be obtained due to the need to preserve vital organs and ACC is prone to vascular nerve invasion and diffuse infiltration, postoperative radiotherapy or radical radiotherapy is required for patients with close resection margins and inoperable patients. For the radiotherapy modality of ACC patients, photon therapy still dominates, but ACC is prone to local recurrence after photon therapy, and the main reasons usually include insufficient irradiation range of the tumor target area, the presence of radiosensitive cell populations in the tumor, or the tumor is in an environment that leads to radiosensitivity (e.g., hypoxia), and so on. Reprogrammed radiation therapy is the most effective radical treatment for patients with locally recurrent ACC, but the efficacy is still unsatisfactory for patients with reprogrammed photon-insensitive tumors, with 2-year LC less than 50% and 5-year LC less than 40% [12]. In recent years, with the advancement of medical equipment, some new radiotherapy techniques, such as proton and carbon ion, have been applied to the clinic. Compared with X-rays, due to the better physical dosimetric advantages of proton and carbon ion beams, especially carbon ions, which are high linear energy transfer (LET) rays with higher relative biological effectiveness (RBE) for tumors that are insensitive to conventional photon therapy [13], they can improve the tumor killing power. Carbon ion beams can energy deposition into Bragg peaks, concentrating the dose in the target tissue, killing tumor cells in the target area while protecting the surrounding and normal tissues on the radiation channel as much as possible to reduce the damage. Because carbon ion has the advantages of good killing effect on cancer cells, short treatment course, no pain and lower side effects, it is especially suitable for the treatment of some solid tumors that are not suitable for surgery, insensitive to conventional rays, and recurring after conventional ray treatment. Therefore, carbon ion is internationally recognized as the most ideal tumor radiotherapy equipment in the 21st century. At present, carbon ion radiotherapy has been clinically applied to the treatment of radiation-resistant tumors of the skull base, head and neck, spine and other bone and soft tissue tumors, pancreatic cancer, gliomas and other refractory tumors, and its therapeutic efficacy is precise [14].
In an early phase I and II clinical study completed by NIRS in Japan [15], carbon ions were used to treat 69 cases of head and neck ACC with a prescribed dose of 57.6-64.0 Gy (RBE) (single split dose of 3.6-4.0 Gy (RBE)/d, 4 times/week), and the target area included only the external radiological safety boundary of the tumor lesion (i.e., no prophylactic irradiation was given), and the 5-year OS, LC, respectively, were 68%, 73%, and no acute grade 4 toxicity was observed, with grade 3 mucositis occurring in 10% of cases and grade 3 skin reactions in 10%.Koto [16] et al. reported the results of 18 cases of locally advanced ACC carbon ion therapy of the tongue root, with a total dose of 57.6-64.0 Gy (RBE) (completed in 16 4-weekly sessions), and 5-year post-treatment LC and OS rates of 92% and 72%, respectively. A retrospective study analyzed 122 patients with head and neck ACC treated with carbon ion radiation therapy, in which the 2-year OS and PFS rates were 94% and 68% [17].Morimoto K et al. reported that 57 patients with unresectable primary ACC who were treated with proton or carbon ion radiation therapy at the Hyogo Prefectural Particle Medical Center from 2003 to 2009 had a 3-year OS of 80% [18 ]. In addition, a study showed that 80 patients who received proton or carbon ion radiation therapy alone between 2002 and 2008 had a 5-year OS of 63% and an LC of 75% [19].In 2004, the University of Heidelberg reported that 21 patients who received photon combined with carbon ion radiation therapy for ACC had a 3-year LC of 62% with no grade 3 or higher toxic side effects [20]. The above findings are consistent with the follow-up results of 5 cases of head and neck ACC treated at our center, which had better local control rates, lower rates of acute toxic reactions and late toxic reactions, and improved local control and overall survival rates with lower toxic side effects after head and neck ACC was treated with carbon ion therapy. For inert tumors like ACC, there may be a higher responsiveness to carbon ion radiation therapy.
At NIRS Japan, Hayashi et al. reported the clinical results of 17 patients with recurrent ACC who were re-treated with carbon ion between 2007 and 2016, and the study demonstrated the feasibility of carbon ion re-programmed radiotherapy for recurrent ACC. The 2-year LC for all patients was 40.5% and the PFS was 29.4%. Of these patients, 37.5% had a grade 3 late toxic response and one had grade 5 CNS necrosis[21]. According to this study, it is suggested that carbon ion therapy for recurrent head and neck ACC has significantly decreased local control rate and progression-free survival compared with the initial treatment, and the toxic side effects are significantly worse, but it can still be used as a salvage therapy.
In addition to the head and neck, cases of ACC originating from the trachea and mammary glands have also been reported. Tracheal adenoid cystic carcinoma (TACC) is a low-grade malignant tumor originating from the mucous glands of the trachea, accounting for 0.04%-0.2% of the total number of malignant tumors in the respiratory system. Shanghai Proton Heavy Ion Hospital 2021 summarized the data of 18 cases of TACC treated with carbon ion therapy, with a median follow-up time of 20.7 months and an overall effective rate of 88.2%, 5 cases developed lung metastasis 12.2-41.0 months after surgery, and 1 case developed local recurrence 31.9 months after carbon ion therapy.The rates of 2-year OS, LC, and PFS were 100%, 100%, and 61.4%. No grade 2-3 toxic reactions were observed, except for one case in which grade 4 tracheal stenosis occurred, which resolved after stent implantation[22]. Adenoid cystic carcinoma of the breast is a small ductal lesion of the breast, which is very rare in the clinic and accounts for 0.1% of all breast cancers; there is also no clear consensus on whether to perform relevant radiotherapy after breast ACC, although ACC located in other sites is good for radiotherapy. However, various studies have shown that breast ACC has a good prognosis, with 5-, 10-, and 15-year OS rates of 98%, 95%, and 91%, respectively; lymph node metastasis rate of <8%, and distant metastasis rate of <20%; and the most common organ for distant metastasis is the lungs, followed by liver, kidney, and bone [23].
Although the observation period of the cases in our center is not enough and the clinical cases are still accumulating, in the past literature reports, for ACC among the various treatments, carbon ion radiotherapy achieved better local control rate and overall survival.The good efficacy of carbon ion radiotherapy for ACC is related to the difference in the biological interactions between carbon ion radiation and photon radiation. The characteristics of highly linear energy transfer (LET) radiation are responsible for the better efficacy of carbon ion efficacy compared to photon radiation, with small changes in cell cycle sensitivity and less or no repair of sub-lethal or potentially lethal cellular damage [24-25].
In summary, carbon ion radiation therapy is an efficacious treatment with low toxicity and side effects for patients with unresectable and postoperative residual, radiotherapy-resistant or recurrent adenoid cystic carcinoma of the head and neck. The five cases of carbon ion therapy for head and neck ACC reported in our center achieved high overall survival and local control rates without grade 3 or higher toxic reactions. The results of a large number of literature review studies suggest that carbon ion therapy can be used as one of the main therapeutic approaches for ACC.
Reference
[1] Coca-Pelaz A, Rodrigo JP, Bradley PJ, Vander Poorten V, Triantafyllou A, Hunt JL, Strojan P, Rinaldo A, Haigentz M Jr, Takes RP, Mondin V, Teymoortash A, Thompson LD, Ferlito A. Adenoid cystic carcinoma of the head and neck--An update. Oral Oncol. 2015 Jul;51(7):652-61. doi: 10.1016/j.oraloncology.2015.04.005. Epub 2015 May 2. PMID: 25943783.
[2] 邱明珠,徐宝卿,汤晓晖,郑智勇,谢艳梅,余英豪,曲利娟.涎腺腺样囊性癌40例临床病理特征及预后分析[J].临床与实验病理学杂志,2021,37(04):428-433.DOI:10.13315/j.cnki.cjcep.2021.04.011.
[3] Unsal A A , Chung S Y , Zhou A H , et al. Sinonasal adenoid cystic carcinoma: a population-based analysis of 694 cases[J]. International Forum of Allergy & Rhinology, 2017.
[4] Sarah A,Odile C,Nicolas F,et al.A prospective multicentre ref-cor study of 470 cases of head and neck adenoid cystic carcinoma:epidemiology and prognostic factors[J].Eur J Cancer,2020,130: 241-249.
[5] 杨子桧. CCL2/CCR2分子轴在涎腺腺样囊性癌嗜神经侵袭中的作用及机制研究[D].中国人民解放军空军军医大学,2019.DOI:10.27002/d.cnki.gsjyu.2019.000024.
[6] Coca-Pelaz A, Rodrigo JP, Bradley PJ, Vander Poorten V, Triantafyllou A, Hunt JL, Strojan P, Rinaldo A, Haigentz M Jr, Takes RP, Mondin V, Teymoortash A, Thompson LD, Ferlito A. Adenoid cystic carcinoma of the head and neck--An update. Oral Oncol. 2015 Jul;51(7):652-61. doi: 10.1016/j.oraloncology.2015.04.005. Epub 2015 May 2. PMID: 25943783.
[7] 李燕,邓文英,李宁,牛萌柯,罗素霞.头颈部腺样囊性癌的临床特征治疗策略和预后[J].中华肿瘤杂志,2019,41(12):932-936.
[8] Rodriguez-Russo CA, Junn JC, Yom SS, Bakst RL. Radiation Therapy for Adenoid Cystic Carcinoma of the Head and Neck. Cancers (Basel). 2021 Dec 17;13(24):6335. doi: 10.3390/cancers13246335. PMID: 34944955; PMCID: PMC8699151.
[9] 杨婧艺,王德辉.鼻腔鼻窦腺样囊性癌治疗进展[J].中国眼耳鼻喉科杂志,2019,19(04):287-292.DOI:10.14166/j.issn.1671-2420.2019.04.019.
[10] Zhang Q, Kong L, Liu R, et al. Chinese ion therapy guideline (Version 2020)[J]. Precision Radiation Oncology,2021,11(3):1-11
[11] Ouyang DQ, Liang LZ, Zheng GS, Ke ZF, Weng DS, Yang WF, Su YX, Liao GQ. Risk factors and prognosis for salivary gland adenoid cystic carcinoma in southern china: a 25~year retrospective study. Medicine (Baltimore). 2017;96:e5964.
[12] Han F,Zhao C,Huang SM,et a1.Long—term outcomes and prognostic factors of re—irradiation for locally recurrent nasopharyngeal carcinoma using intensity—modulated radiotherapy[J].Clin Oncol(R CoIl Radi01),2012,24(8):569—76.
[13] Jiikel O Medical physics aspects of particle therapy[J].Radiat ProtDosimetry,2009,137(1—2):156
[14] Kamada T'Tsujii H,Blakely EA,et a1.Carbon ion radiotherapy in japan:An assessment of20 years ofclinical experience[J].Lancet Oncol,2015,16(2):e93一e100.
[15] Mizoe JE,Tsujii H,Kamada T,et a1.Dose escalation study ofcarbon ion radiotherapy for locally advanced head~~and·~neck cancer[J].Inl J Radiat OncoJ Biol Phys,2004,60(2):358·364.DOI:10.1016/j.ijrobp.2004.02.067.
[16] Koto M,Hasegawa A,Takagi R,et a1.Evaluation of the safety and efficacy of carbon ion radiotherapy for locally advanced adenoid cystic carcinoma of the tongue base[J].Head Neck,2016.DOI:lO.1002/hed.24397.
[17] Sulaiman NS, Demizu Y, Koto M, Saitoh JI, Suefuji H, Tsuji H, et al. Multicenter study of carbon-ion radiation therapy for adenoid cystic carcinoma of the head and neck: subanalysis of the Japan Carbon-Ion Radiation Oncology Study Group (J-CROS) Study (1402 HN). Int J Radiat Oncol Biol Phys. (2018) 100:639–46. doi: 10.1016/j.ijrobp.2017.11.010
[18] Morimoto K, Demizu Y, Hashimoto N, Mima M, Terashima K, Fujii O, Otsuki N, Murakami M, Fuwa N, Nibu K. Particle radiotherapy using protons or carbon ions for unresectable locally advanced head and neck cancers with skull base invasion. Jpn J Clin Oncol. 2014 May;44(5):428-34. doi: 10.1093/jjco/hyu010. Epub 2014 Mar 11. PMID: 24620027.
[19] Takagi M, Demizu Y, Hashimoto N, Mima M, Terashima K, Fujii O, Jin D, Niwa Y, Morimoto K, Akagi T, Daimon T, Sasaki R, Hishikawa Y, Abe M, Murakami M, Fuwa N. Treatment outcomes of particle radiotherapy using protons or carbon ions as a single~modality therapy for adenoid cystic carcinoma of the head and neck. Radiother Oncol. 2014;113:364–70.
[20] Schulz~Ertner D, Nikoghosyan A, Thilmann C, Haberer T, Jakel O, Karger C, Kraft G, Wannenmacher M, Debus J. Results of carbon ion radiotherapy in 152 patients. Int J Radiat Oncol Biol Phys. 2004;58:631–40.
[21] Hayashi K, Koto M, Ikawa H, Hagiwara Y, Tsuji H, Ogawa K, Kamada T. Feasibility of re~irradiation using carbon ions for recurrent head and neck malignancies after carbon~ion radiotherapy. Radiother Oncol. 2019;136:148–53.
[22] Chen J, Mao J, Ma N, Wu KL, Lu J, Jiang GL. Definitive carbon ion radiotherapy for tracheobronchial adenoid cystic carcinoma: a preliminary report. BMC Cancer. 2021 Jun 26;21(1):734. doi: 10.1186/s12885-021-08493-1. PMID: 34174854; PMCID: PMC8236132.
[23] Ghabach B, Anderson WF, Curtis RE, et al. Adenoid cystic carcinoma of the breast in the United States (1977 to 2006): a population-based cohort study[J]. Breast Cancer Res,2010,12(4):R54.
[24] Pommier P, Liebsch NJ, Deschler DG, Lin DT, McIntyre JF, Barker FG II, Adams JA, Lopes VV, Varvares M, Loeffler JS, Chan AW. Proton beam radiation therapy for skull base adenoid cystic carcinoma. Arch Otolaryngol Head Neck Surg. 2006;132:1242–9.
[25] Kanai T, Matsufuji N, Miyamoto T, Mizoe J, Kamada T, Tsuji H, Kato H, Baba M and Tsujii H: Examination of GyE system for HIMAC carbon therapy. Int J Radiat Oncol Biol Phys 64: 650- 656, 2006. PMID: 16414376. DOI: 10.1016/j.ijrobp.2005.09.043