Preliminary Efficacy Observation of Carbon Ion Radiotherapy in the Treatment of Muscle-invasive Blad
Preliminary Efficacy Observation of Carbon Ion Radiotherapy in the Treatment of Muscle-invasive Bladder Cancer in Phase I/II Clinical Trials
中华放射医学与防护杂志 2022年 9月第 42 卷第9期 ChinJ Radiol Med Prot, September 2022,Vol. 42,No. 9
Zhang Yanshan, Li Xiaojun,Zhang Yihe,Pan Xin, Wang Xin,Yang Yuling,Chai Hongyu,Ma Tong
Heavy Ion Radiotherapy Department, Wuwei Cancer Hospital of Gansu Province,Wuwei 733000,China
Corresponding author: Li Xiaojun, Email: anglwe@sina.com
[Abstract] Objective :To evaluate the feasibility and safety of carbon ion radiotherapy(CIRT) in the treatment of muscle-invasive bladder cancer in phase Ⅰ/Ⅱ clinical trials.Methods Clinical stage T2.3 patients with muscle-invasive bladder cancer ( without distant metastasis) were studied. A three-fraction treatment was applied, including the local irradiation with the dose from 12 Gy to 24 Gy and 11 fractions of whole-bladder irradiation with a dose of 44 Gy. The carbon ion irradiation dose is determined with relative biological effectiveness(RBE) of 3.0. The total dose for bladder tumor was56-68 Gy in 14 fractions. The primary endpoints included tumor treatment-related side effects, dose-limiting toxicity (DLT) responses, and local control(LC) rate,and the secondary endpoints included progression-free survival(PFS). Results Nine patients receivedCIRT of various doses in the clinical trials,with the dose gradually increasing to 68 Gy. The patients did not suffer from DLT response, acute adverse effects of radiation therapy of grade ≥3, and late radiation adverse reactions during follow-up. When the dose to the tumor reached 68 Gy, there were 2 cases of grade 2 acute urogenital tract reaction and 1 case of acute lower gastrointestinal tract symptom. For the group with a dose above 62 Gy, three cases of grade 1 late radiation bladder reaction were observed and their symptoms included urinary frequeney and microscopic hematuria. At the end of treatment, hematuria disappeared,dysuria was relieved, and urine red blood cell value significantly decreased for all the patients. Three months and six months after treatment, the LC rates were 100% and 88. 9%, respectively, and the objective response rates were both 88.9%. One patient developed local recurrence and was treated with salvage surgery six months after treatment. Conclusions The preliminary efficacy observation of CIRT in the treatment of muscle-invasive bladder cancer showed significant short-term efficacy,obvious symptom relief, and good tolerability for patients,without DLT. Therefore,CIRT is safe and feasible.
[Key words] Carbon ion radiotherapy; Muscle invasive bladder cancer(MIBC);Clinical trial; Efficacy observation; Adverse reactions
Fund programs: Key R&D Plan of Science and Technology Program of Gansu Province
(19YF3FH001);The Innovation Baseand Talent Program of Science and Technology Department of Gansu Province (21JR7RH896)
Trial registration: Chinese Clinical Trial Registry,ChiCTR 2100043607
DOI :10.3760/cma.j.cn 112271-20220508-00197
For localized muscle-invasive bladder cancer, the National Comprehensive Cancer Network (NCCN) recommends maximal transurethral resection of bladder tumor combined with concurrent chemoradiotherapy as an alternative to radical cystectomy [1]. For patients with muscle-invasive bladder cancer (MIBC) who are unable to tolerate chemotherapy or are unsuitable for or refuse cystectomy, radiotherapy is a conventional treatment option. Compared to photon beams, carbon ions offer both physical and biological advantages. Whether carbon ion radiotherapy can improve outcomes in MIBC is worth exploring. The primary reason other international carbon ion therapy centers have not conducted research on bladder cancer is the challenge of target displacement due to changes in bladder volume and tension during carbon ion therapy for bladder tumors. The Wuwei Heavy Ion Center has innovatively developed the "Galloping Scheme" [2], which addresses the issue of bladder volume and positional changes during radiotherapy, ensuring precision in treatment delivery. Therefore, our center has initiated a prospective phase I/II clinical study on carbon ion radiotherapy for MIBC.
Materials and Methods
- Case Data: Nine patients with muscle-invasive bladder cancer (MIBC) were prospectively selected.
Inclusion Criteria:
- Diagnosis of MIBC confirmed by cystoscopy and biopsy.
- Clinical stage T2~3.
- Eastern Cooperative Oncology Group (ECOG) performance status score of 0~1.
- Expected survival ≥12 months.
Exclusion Criteria: - No prior radiotherapy.
- Refusal to undergo radical cystectomy.
- Comorbidities unsuitable for radical cystectomy.
- No diffuse metastasis.
- No bilateral ureteral obstruction.
The study was approved by the Institutional Ethics Committee (Approval No.: 2020-Ethical Review-12), and written informed consent was obtained from all patients.
2. Study Design of Carbon Ion Radiotherapy for Muscle-Invasive Bladder Cancer: The Wuwei Heavy Ion Center (WWHIC) conducted multiple phase I/II dose-escalation studies targeting malignancies at different sites. This study focused on dose escalation for carbon ion radiotherapy in bladder cancer. The flowchart is shown in Figure 1.
• Phase I: The maximum safe dose of carbon ion radiotherapy for muscle-invasive bladder cancer was determined. The trial followed the "3+3" principle: if only 1 out of 6 patients experienced dose-limiting toxicity (DLT), the dose was escalated; if 2 or more patients experienced DLT, the study was terminated, and the previous dose level was defined as the prescription dose.
• Phase II: Based on literature review, the 2-year local control rate for bladder cancer with conventional radiotherapy is 41%~54%[3] . The expected 2-year local control rate with carbon ion radiotherapy is 80%. The study aimed to observe whether the 2-year local control rate in enrolled patients was superior to photon radiotherapy, as well as long-term efficacy and late adverse effects.
Primary Endpoints: Treatment-related adverse reactions, DLT responses, and local control rate.
Secondary Endpoint: Progression-free survival (PFS).
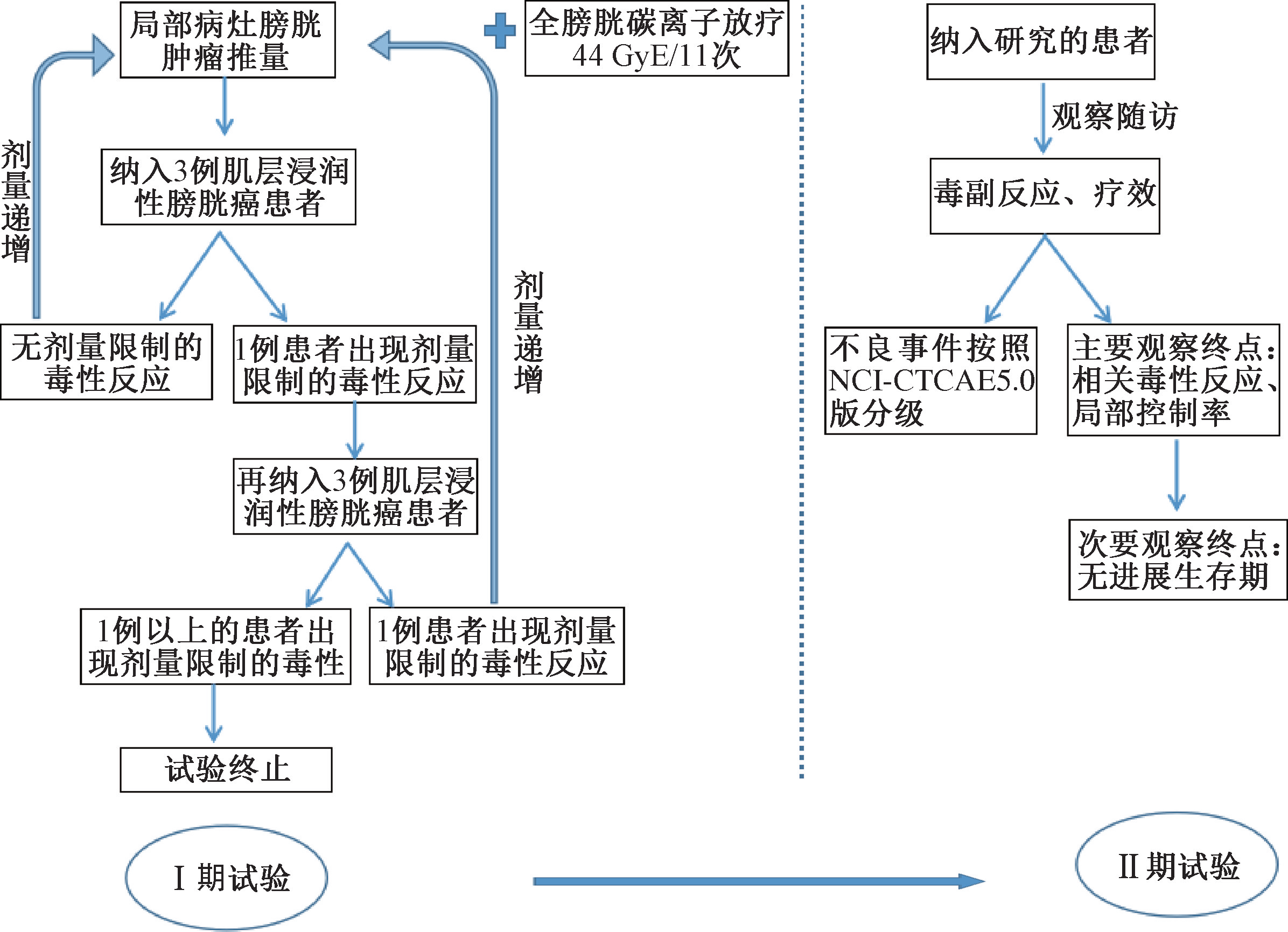
Figure 1: Flowchart of Phase I/II Clinical Trials of Carbon Ion Radiotherapy of Muscle-Invasive Bladder Cancer.
3. Carbon Ion Radiotherapy Plan
CT localization was performed using the Siemens SOMATOM Confidence CT with a large aperture. Patients were positioned in the supine position, with the rectum emptied before localization[2], and fixed using a pelvic vacuum cushion and thermoplastic film. The "Galloping Scheme" was employed to maintain stable bladder volume and position 2. During carbon ion therapy, bladder volume-pressure control technology was combined, with a urinary catheter inserted and connected to a water-sealed bottle at atmospheric pressure. Based on the principle of the communicating vessel, the actual bladder volume was adjusted and maintained according to the actual situation. The pressure of the water-sealed bottle was adjusted to keep the bladder volume and pressure constant, ensuring that the bladder volume remained stable during carbon ion radiotherapy (CIRT). The CT localization scan ranged from the ischial tuberosity to the L5 level, with a slice thickness of 3 mm. Treatment planning was performed on non-contrast CT images. The target area was delineated using the Varian Eclipse 15.5 radiotherapy planning system (Vital Beam accelerator, Varian Medical Systems, USA), and the treatment plan was designed using the ciPlan carbon ion radiotherapy planning system (TPS, version 1.0, Lanzhou Kejin Taiji New Technology Co., Ltd.). Target delineation followed the principles of radiation oncology theory and practice. The gross tumor volume (GTV) was defined as the bladder lesion visible on CT or positron emission tomography CT images. The planning target volume 1 (PTV1) was the boost target area for the local lesion, with a 0.5 cm margin added to the GTV. The clinical target volume (CTV) included the entire bladder, and the internal target volume (ITV) was uniformly expanded by 0-3 cm around the bladder. To eliminate setup errors and dose distribution uncertainties, the planning target volume 2 (PTV2) was delineated with a 3-6 mm margin added to the ITV. Considering tumor volume changes during treatment, a local boost was first given to the primary tumor site, followed by sequential whole-bladder radiotherapy. On the planning CT, bilateral femoral heads, small intestine, colon, and rectum were delineated. The delineation of organs at risk followed the American Radiation Therapy Oncology Group (RTOG) recommendations for pelvic normal tissue delineation[4]. Dose constraints were based on the Chinese Ion Therapy Guidelines[5]: small intestine: V58 < 0-0.03 ml, V50 < 10 ml, V45 < 30 ml; bladder: V65 < 25%; femoral heads: V45 < 40%, Dmax < 50 Gy; rectum: Dmean < 50 Gy, Dmax < 66 Gy, D5 < 60 Gy, D10 < 50 Gy.
The purpose of this trial was to explore the maximum tolerated dose, determine the optimal dose, and observe efficacy and safety. Phase I focused on safety evaluation to identify the maximum tolerated dose (optimal dose), while Phase II focused on both safety and efficacy evaluation. In this protocol, the dose for PTV1 was fixed at 44 Gy in 11 fractions, with 4 Gy per fraction, and PTV2 was treated in 3 fractions. The patient cohort included at least 3 patients at each dose level. Based on discussions by the research team every 6 months, dose escalation was performed after carefully observing acute radiotherapy adverse reactions in normal tissues. The total dose for bladder tumors was 56-68 Gy, with sequential radiotherapy of PTV1 and PTV2, totaling 14 fractions. Based on previous carbon ion therapy experience, the clinical dose range for other tumors was 51.2-72 Gy[6]. After reaching the maximum dose of 68 Gy, no further dose escalation was performed. No other anti-tumor combination therapies were administered during the Phase I/II trials.
4. Adverse Reactions and Efficacy Evaluation
Before treatment, patients underwent physical examinations, complete blood count, urinalysis, comprehensive biochemical tests, pelvic MRI, and 18F-PETCT. During treatment, complete blood count, urinalysis, comprehensive biochemical tests, physical examinations, and adverse reaction assessments were performed at least once a week. At the end of treatment, pelvic MRI was performed. After CIRT completion, follow-ups were conducted every 1-3 months for 5 years. Acute and late adverse reactions during carbon ion therapy and within the 5-year follow-up period were observed. Adverse reactions were evaluated based on clinical symptoms, signs, and test results, following the Common Terminology Criteria for Adverse Events version 5.0 (CTCAE v5.0) and the European Organization for Research and Treatment of Cancer (EORTC)/RTOG late radiation injury grading criteria. Dose-limiting toxicity (DLT) was defined as grade 3 or higher genitourinary or lower gastrointestinal adverse events occurring within 24 weeks of radiotherapy initiation, as evaluated by CTCAE v5.0, and late bladder or lower gastrointestinal radiation reactions as evaluated by the EORTC/RTOG late radiation injury grading criteria. Tumor efficacy was evaluated according to the Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1). Short-term efficacy evaluation indicators included 3- and 6-month local control rates and objective response rates. Long-term efficacy evaluation included progression-free survival and overall survival after treatment.
5. Statistical Analysis
This study was designed as a single-group target value study. Statistical analysis was performed using SAS 9.4 software. In addition to necessary descriptive statistics and 95% confidence interval estimation for general observation or evaluation indicators, a hypothesis test with a 60% target value and a two-sided significance level of 0.05 was used. Data conforming to a normal distribution were described as xˉ±s, and results were analyzed using two-sided t-tests or non-parametric tests. A P-value < 0.05 was considered statistically significant.
Results
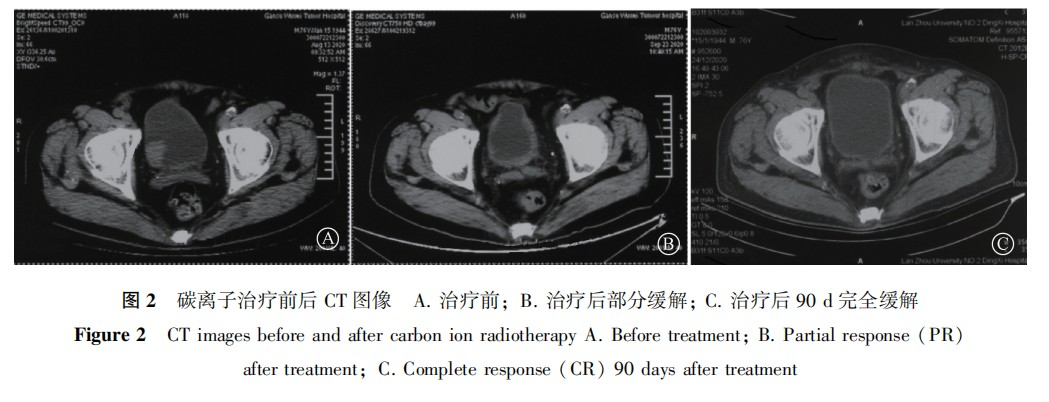
- Patient Data: From March 2020 to January 2022, nine patients were enrolled in the Phase I trial. The baseline clinical characteristics of the patients are shown in Table 1. The median age was 71 years, with 8 cases classified as stage II and 1 case as stage III. The median tumor size was 4.5 cm (range: 3.0–7.8 cm). In terms of pathological types, except for one rare case of urachal mucinous adenocarcinoma, the rest were urothelial carcinomas. Regarding tumor locations, two cases had multiple bladder nodules, including one with nodules in the bladder trigone and posterior wall, and one with lesions in the urachus and bladder dome. All nine patients successfully completed three dose levels of carbon ion therapy, with a median follow-up time of 13 months. Figure 2 shows the comparison of imaging data before treatment, after treatment, and 90 days after treatment in one bladder cancer patient.
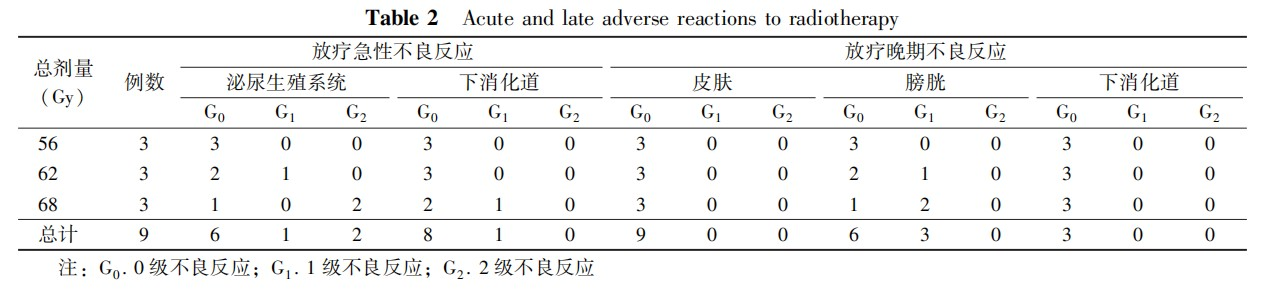
- Acute and Late Adverse Reactions to Radiotherapy: All observed acute and late adverse reactions are listed in Table 2. Although three patients experienced acute genitourinary (GU) adverse reactions (grade 1–2) and one patient experienced lower gastrointestinal symptoms (grade 1), all patients completed the planned treatment. Acute reactions mostly occurred during treatment and within two weeks after treatment, manifesting as radiation cystitis, urethritis, and proctitis. A grade 2 acute genitourinary reaction was observed at a tumor dose of 68 Gy. Due to the significant dose reduction before and after the Bragg peak of the carbon ion beam and the deep location of the bladder irradiation area, no radiation skin reactions were observed. For late adverse reactions, three cases of grade 1 radiation cystitis were observed in the 62 Gy and 68 Gy dose groups, occurring more than three months after radiotherapy, presenting as urinary frequency and microscopic hematuria. No lower gastrointestinal reactions of any grade were observed. No dose-limiting toxicity (DLT) occurred in any of the nine patients.
3. Efficacy Evaluation After Carbon Ion Therapy: At the end of carbon ion therapy across all dose levels, hematuria disappeared, dysuria was alleviated, and the urinary red blood cell count significantly decreased from an average of 19,673 cells/μl to an average of 92 cells/μl. At 3 months post-treatment, 1 case achieved complete remission, 7 cases achieved partial remission, the local control rate was 100%, and the objective response rate was 8/9. At 6 months post-treatment, 4 cases achieved complete remission, 4 cases achieved partial remission, the local control rate was 8/9, and the objective response rate was 8/9. One patient experienced local recurrence within the 6-month follow-up period and underwent salvage total cystectomy.
Discussion
The primary treatment for muscle-invasive bladder cancer (MIBC) is radical cystectomy (RC) combined with pelvic lymph node dissection (PLND). However, the perioperative complication rate of radical cystectomy ranges from 28% to 64%, with a mortality rate of 2.5% to 2.7%. The main causes of death include cardiovascular complications, sepsis, pulmonary embolism, liver failure, and massive hemorrhage. The number and proportion of elderly bladder cancer patients are continuously increasing. In 2015, Comploj et al[7]. retrospectively studied 251 elderly MIBC patients who underwent radical cystectomy. The 30-day and 90-day mortality rates for patients aged 75 and above were 11.0% and 45.8%, respectively. Radical cystectomy in elderly MIBC patients does not provide satisfactory survival benefits and significantly reduces quality of life.
A meta-analysis including 12 studies confirmed that bladder-preserving comprehensive treatment based on radiotherapy can achieve survival rates comparable to surgery. The 5-year survival rate after bladder preservation is 45.5% to 63.7%, with no significant difference in 5-year and 10-year survival rates or 5-year distant metastasis rates compared to radical cystectomy[8]. For most MIBC patients who wish to preserve their bladder, maximal transurethral resection of bladder tumor (TURBT) followed by cisplatin-based chemotherapy combined with radiotherapy can achieve complete remission rates of 60% to 80% and 5-year survival rates of 50% to 60%, similar to radical cystectomy. However, for elderly patients or those with comorbidities who cannot tolerate surgery or chemotherapy, radical radiotherapy alone, even with a prescription dose increased to 64–70 Gy, still results in tumor residue in 50% to 60% of patients based on histopathological examination. The 5-year survival rate with radiotherapy alone is only 20% to 40%, and the 5-year local control rate is only 40%. Radiotherapy alone is less effective and often requires combined chemotherapy, surgery, and other comprehensive treatments to preserve bladder function[9].
Compared to photon beams, carbon ion beams deposit more energy at the end of their radiation range, forming a Bragg peak while reducing radiation damage to surrounding tissues. Carbon ion radiotherapy (CIRT) causes double-strand breaks in the DNA of 70% of tumor cells, which are difficult to repair, leading to tumor cell death. Additionally, CIRT sensitivity is independent of the cell cycle, with a high relative biological effectiveness (RBE) and low oxygen enhancement ratio. This radiation also causes double-strand breaks in DNA molecules, inflicting fatal damage to tumor cells[10⁃11].
Due to the different RBE models used by major carbon ion therapy centers internationally, a standardized treatment protocol and guidelines have not yet been established. Initially, our center’s prescription dose and fractionation were based on the dose fractionation scheme required by the National Medical Products Administration for the pre-market safety and efficacy evaluation of the carbon ion therapy system. In August 2020, the first bladder cancer patient was treated with a prescription dose of 64 Gy, based on the pelvic tumor irradiation dose during the clinical trial, with good results and no short-term adverse reactions[12]. For newly treated diseases, dose escalation trials are necessary. Currently, the dose conversion between carbon ion and photon therapy requires further research. The initial dose escalation is consistent with photon doses, and the prescription dose is determined through safety evaluation. The Phase I/II clinical trial is the first dose escalation study of CIRT for bladder cancer. The recommended dose for photon radical radiotherapy is 60–66 Gy, with a large field irradiation of 40–45 Gy followed by a boost to the bladder or tumor. In the Phase I trial design, the dose for the whole bladder target volume (PTV2) was 44 Gy, consistent with photon radiotherapy, while the total dose for the bladder tumor target volume (PTV1) was 56–68 Gy, based on the photon radical dose and the normal tissue dose limits for the bladder in RTOG 0415.
In this study, a grade 2 acute genitourinary reaction was observed in the 68 Gy dose group, presenting with bladder irritation symptoms such as urinary frequency, dysuria, and turbid fibrous floccules in the urine, which were caused by focal exfoliation of the transitional epithelium in acute radiation cystitis. These symptoms persisted for two weeks after radiotherapy and then resolved. Grade 1 gastrointestinal reactions, such as frequent bowel movements and tenesmus, usually required no intervention. Late radiation adverse reactions included only chronic radiation cystitis, with nocturia frequency doubling compared to pre-treatment levels and microscopic hematuria caused by mild capillary dilation. The main causes were radiation-induced vascular endothelial cell swelling and necrosis, gradually developing into persistent inflammatory cell infiltration, combined with epithelial hyperplasia, vascular changes, fibrous exudation, lamina propria edema, hyalinization, and capillary dilation in the bladder mucosa. The bladder doses for the three cases of radiation cystitis were Dmax 62.2, 68.9, and 68.4 Gy, with V62 21.3%, V68 18.4%, and 9.8%, respectively. A retrospective analysis by Shanghai Proton and Heavy Ion Center of 154 prostate cancer patients treated with CIRT showed that when V61.5 > 1.7% and Dmax > 63 Gy, the probability of grade 1 and 2 acute bladder adverse reactions was 37.0%, with a 15.6% [13]probability of grade 2 reactions. The bladder reactions and doses observed in this study were consistent with these findings.
Currently, other particle centers have only conducted CIRT for pelvic tumors involving the bladder. Miyasaka et al[14]. reported the treatment of 7 patients with locally advanced (cT4) prostate cancer invading the bladder using CIRT. The involved bladder wall received 57.6 Gy in 16 fractions, and with a median follow-up of 78 months, only 1 case of grade 2 acute urinary reaction and 1 case of grade 2 late adverse reaction (hematuria and urgency) were observed. The Phase I/II dose escalation study 9702 [15]conducted by the National Institute of Radiological Sciences in Chiba, Japan, included 13 patients with bladder involvement. They received 68.8–72.8 Gy, with only 1 case of grade 1 acute urinary reaction and 1 and 5 cases of grade 1 and 2 late adverse reactions, respectively. These results indicate that CIRT is safe for bladder tumors.
In this clinical study, 1 case of disease progression (PD) occurred within the 6-month follow-up period after treatment, and salvage total cystectomy was performed. The recurrence was analyzed to be due to the tumor’s location in the bladder trigone, with no dose-limiting conditions such as intestinal involvement and good reproducibility of treatment positioning. The dose gradient was in the second dose segment of 62 Gy. Tumor radiation tolerance and resistance are often important reasons for recurrence in some tumors. Carbon ions are high-LET radiation, independent of the oxygen environment around tumor cells, with minimal sublethal damage repair. It is considered whether certain gene or protein expression abnormalities may lead to CIRT resistance, resulting in tumor recurrence.
A challenging issue in bladder cancer radiotherapy is how to account for changes in bladder volume and tumor position during treatment to ensure target accuracy. Using this technology, bladder volume changes before each treatment remain constant, with a standard deviation of less than ±5% during treatment. The internal target volume (ITV) changes in the bladder averaged 0.1–0.3 cm, significantly lower than the 1.4–2.0 cm [16]reported in similar studies. The dose distribution in the carbon ion peak region matched the initial plan. The dose tolerance changes for normal pelvic tissues (small intestine, colon, and rectum) were within ±5%. Currently, a bladder cancer treatment pressure measurement device has been patented (Patent No.: CN215348989).
The limitations of this study include the inability to evaluate the correlation between local tumor response and dose escalation in the dose escalation study, as the 6-month objective response rate after CIRT was 88.9%, and the local recurrence case was in the 62 Gy dose segment. Future Phase II trials will expand the sample size to observe and evaluate the correlation between local tumor response and dose within 2 years.
In conclusion, the preliminary efficacy observation of Phase I/II clinical trials of CIRT for muscle-invasive bladder cancer shows that CIRT is safe and feasible, with no dose-limiting toxicity, significant short-term efficacy, obvious symptom relief, and good patient tolerance. Compared to RC, CIRT not only achieves similar survival outcomes but also reduces complication rates, thereby improving quality of life.
Conflict of Interest: None.
Author Contributions: Zhang Yanshan was responsible for designing the research plan; Li Xiaojun was responsible for implementing the research, writing, and revising the manuscript; Zhang Yihe participated in manuscript revision; Pan Xin, Wang Xin, Yang Yuling, and Chai Hongyu participated in data collection; Ma Tong was responsible for manuscript review.
Reference
[1] National Comprehensive Cancer Network. The NCCN bladder cancer clinical practice guidelines in oncology (version 4, 2020) [EB/OL]. Fort Washington: NCCN, 2020 [2020-04-28]. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp.
[2] Zhang YS, Li XJ, Zhang YH, et al. Carbon ion radiotherapy for bladder cancer: a case report [J]. World J Clin Cases, 2021, 9(26): 7833–7839. DOI: 10.12998/wjcc.v9.i26.7833.
[3] Coppin CM, Gospodarowicz MK, James K, et al. Improved local control of invasive bladder cancer by concurrent cisplatin and preoperative or definitive radiation. The National Cancer Institute of Canada Clinical Trials Group [J]. J Clin Oncol, 1996, 14(11): 2901–2907. DOI: 10.1200/JCO.1996.14.11.2901.
[4] Gay HA, Barthold HJ, O’Meara E, et al. Pelvic normal tissue contouring guidelines for radiation therapy: a Radiation Therapy Oncology Group consensus panel atlas [J]. Int J Radiat Oncol Biol Phys, 2012, 83(3): 353–362. DOI: 10.1016/j.ijrobp.2012.01.023.
[5] Zhang Q, Kong L, Liu R, et al. Chinese ion therapy guideline (Version 2020) [J]. Pre Radiat Oncol, 2021, 11(3): 1–11. DOI: 10.1002/pro6.1120.
[6] Kamada T, Tsujii H, Blakely EA, et al. Carbon ion radiotherapy in Japan: an assessment of 20 years of clinical experience [J]. Lancet Oncol, 2015, 16(2): 93–100. DOI: 10.1016/S1470-2045(14)70412-7.
[7] Comploj E, West J, Mian M, et al. Comparison of complications from radical cystectomy between old-old versus oldest-old patients [J]. Urol Int, 2015, 94(1): 25–30. DOI: 10.1159/000358731.
[8] Zhao J, Zhou L, Pan Y, et al. A systematic review and meta-analysis of radical cystectomy in the treatment of muscular invasive bladder cancer (MIBC) [J]. Transl Androl Urol, 2021, 10(8): 3476–3485. DOI: 10.21037/tau-21-564.
[9] Sauer R, Birkenhake S, Kühn R, et al. Efficacy of radiochemotherapy with platin derivatives compared to radiotherapy alone in organ-sparing treatment of bladder cancer [J]. Int J Radiat Oncol Biol Phys, 1998, 40(1): 121–127. DOI: 10.1016/s0360-3016(97)00579-8.
[10] Kanai T, Endo M, Minohara S, et al. Biophysical characteristics of HIMAC clinical irradiation system for heavy-ion radiation therapy [J]. Int J Radiat Oncol Biol Phys, 1999, 44(1): 201–210. DOI: 10.1016/S0360-3016(98)00544-6.
[11] Kanai T, Matsufuji N, Miyamoto T, et al. Examination of GyE system for HIMAC carbon therapy [J]. Int J Radiat Oncol Biol Phys, 2006, 64(2): 650–656. DOI: 10.1016/j.ijrobp.2005.09.043.
[12] Lee WR, Dignam JJ, Amin M, et al. NRG Oncology RTOG 0415: A randomized phase III non-inferiority study comparing two fractionation schedules in patients with low-risk prostate cancer [J]. Int J Radiat Oncol Biol Phys, 2016, 94(1): 3–4. DOI: 10.1016/j.ijrobp.2015.10.049.
[13] Li Y, Li P, Hsi W, et al. Normal tissue complication probability (NTCP) models of acute urinary toxicity (AUT) following carbon ion radiotherapy (CIRT) for prostate cancer [J]. Radiother Oncol, 2021, 156: 69–79. DOI: 10.1016/j.radonc.2020.12.009.
[14] Miyasaka Y, Kawamura H, Sato H, et al. Carbon ion radiotherapy for prostate cancer with bladder invasion [J]. BMC Urol, 2021, 21(1): 106. DOI: 10.1186/s12894-021-00871-y.
[15] Kato S, Ohno T, Tsujii H, et al. Working Group of the Gynecological Tumor. Dose escalation study of carbon ion radiotherapy for locally advanced carcinoma of the uterine cervix [J]. Int J Radiat Oncol Biol Phys, 2006, 65(2): 388–397. DOI: 10.1016/j.ijrobp.2005.12.050.
[16] Kochan A, Rives R, Galloway K, et al. Targeting the tumor: assessing the impact of bladder volume and position on accuracy of radiation delivery for patients with bladder cancer [J]. Cureus, 2017, 9(9): 1638. DOI: 10.7759/cureus.1638.
