Carbon Ion Therapy for Recurrent Lower Leg Myxoid Liposarcoma
Carbon ion radiotherapy for recurrence of calf myxoid liposarcoma after the second postoperative radiotherapy: A case report
LI Xiaojun1,ZHANG Yanshan1,YE Yancheng1,CHEN Dongji1,Ll Sha2,PAN Xin1
1Heavy lon Radiotherapy Department, Wuwei Tumor Hospital of Gansu Province ,Gansu Wuwei 733000 ,China; 2Depariment of Radiotherapy , the 940th Hospital of Joint Logistics Support Force of Chinese People's Liberation Army, Gansu Lanzhou 730000, China.
【Key words 】carbon ion radiotherapy, carbon ion therapy system ,soft tissue sarcoma, curative effect
Modem 0ncology 2022, 30(02):0314 -0318
【Chinese Library Classification】R738.6 【Document Code】B DOI: 10.3969/j.issn.1672-4992.2022.02.029
【Article ID】1672-4992(2022)02-0314-05
The incidence of soft tissue sarcoma is 1.28-1.72 per 100,000 people, with 50%-60% occurring in the limbs, among which liposarcoma accounts for 25%-30% of all soft tissue sarcomas. Myxoid liposarcoma is the second most common subtype of liposarcoma, representing 25%-50% of all liposarcomas [1]. According to the 2015 edition of the "Chinese Expert Consensus on Diagnosis and Treatment of Soft Tissue Sarcomas," secondary surgical resection for locally recurrent soft tissue sarcomas is classified as a Grade 1 expert-recommended consensus. The recommended surgical extent involves removing the recurrent tumor and scar tissue, along with >5 cm of surrounding normal tissue. For cases where preoperative antitumor therapy fails to enable surgical resection, or where surgery may severely impair limb function, or where multiple surgeries would cause extensive damage, and radiotherapy is the preferred option, high-LET particle irradiation using protons or heavy ions can be employed [2]. Heavy ions generally refer to charged ions with an atomic number (Z) >2. In clinical treatment, the heavy ion beams used typically refer to carbon ion beams. The advancement of heavy ion radiotherapy is reflected in both radiophysical and radiobiological effects. Radiophysically, the dose of heavy ions is primarily deposited at the end of their range (Bragg peak), significantly reducing the radiation exposure to normal tissues and thereby minimizing radiotoxic side effects. Radiobiologically, carbon ions exhibit a high relative biological effectiveness (RBE), inducing DNA double-strand breaks that are difficult to repair. Soft tissue sarcomas are resistant to conventional X-ray therapy and respond poorly to treatment. Clinical trial results from the National Institute of Radiological Sciences (NIRS) in Japan demonstrated that carbon ion therapy for soft tissue sarcomas achieved a 5-year local control rate of 69%-79%, a 3-year survival rate of 47%-68%, and a 5-year survival rate of 36%-52% [3-5], showing significant therapeutic efficacy. China is the fourth country in the world to conduct heavy ion therapy for tumors and related experimental research. The first domestically developed carbon ion therapy device (HIMM), created by the Institute of Modern Physics of the Chinese Academy of Sciences, was installed at the Heavy Ion Center of Wuwei Cancer Hospital in Gansu Province. From November 6, 2018, to May 23, 2019, a clinical trial was conducted to verify its safety and efficacy. The trial included one patient with recurrent myxoid liposarcoma in the right calf following secondary surgery and radiotherapy, who achieved excellent therapeutic outcomes. The case is reported as follows.
1 Case Report
A 55-year-old female patient was admitted with the chief complaint of "more than two years after secondary surgery and radiotherapy for myxoid liposarcoma of the right calf". The patient underwent resection of a huge mass in the right calf due to a "mass in the right calf" on November 17, 2014. Postoperative pathological examination showed: (Right calf) The examined tumor tissue exhibited a lobulated structure, with a tumor size of 9.6 cm × 4 cm × 5 cm. The tumor cells were composed of uniform oval and short spindle-shaped primitive mesenchymal cells mixed with signet-ring-like lipoblasts. The stroma was myxoid, with branching thin-walled capillaries and visible mucin lake formation. Focally, the tumor cells were round and arranged in a sheet-like pattern (<10%). The lesion was diagnosed as myxoid liposarcoma. IHC: S-100 (-), Ki-67 (2% - 5% +). On March 30, 2016, a follow-up right lower limb MRI showed postoperative changes of right calf liposarcoma, with multiple nodular shadows in the right muscle interspace, considered as recurrence. Therefore, on April 7, 2016, under general anesthesia, the patient underwent resection of the recurrent lesion and radiofrequency ablation for postoperative recurrence of right calf liposarcoma. The postoperative pathology showed myxoid liposarcoma. In May 2016, the patient received postoperative radiotherapy, with an initial course of DT 50.4 Gy/28f and a subsequent course of reduced-field boost of DT 10 Gy/5f. On November 1, 2018, a follow-up PET-CT (Figure 1A) showed postoperative changes of myxoid liposarcoma in the right calf, with disordered surgical area structure. PET revealed mild small-patchy increased radioactive uptake, with a size of approximately 15 mm × 13 mm × 19 mm, considered as a malignant tumor recurrence. The right lower limb MRI (Figure 1B) showed a nodular isointense T1 and slightly hyperintense T2 signal shadow in the middle interstice of the right tibia and fibula, with a size of approximately 4.2 cm × 2 cm. The internal signal was uneven, and there were a few streaky exudative signals around it. Considering the medical history, it was mostly considered as postoperative recurrence of liposarcoma. The clinical diagnosis was recurrence of myxoid liposarcoma of the right calf after surgery and radiotherapy, pT2bN0M0G3 stage III, rpT1bN0M0G3 stage IIA. ECOG score: 1 point. This study met the ethical standards set by the clinical trial ethics committee of our hospital, with an ethics review number of 2018 - Ethics Review - 11. This patient had complete clinical data and had provided informed consent.
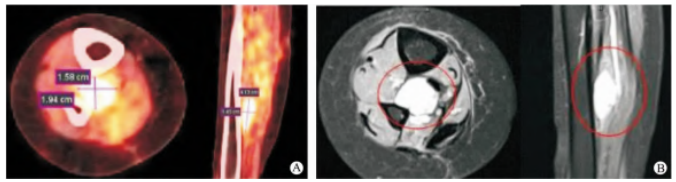
Fig. 1 The patient's re-examination results on November 1,2018
A: PET-CT examination result B: MRI examination result in right lower extremity.
After comprehensive examination and evaluation, the patient was enrolled in the "Validation of Safety and Efficacy of Carbon Ion Therapy System" clinical trial in November 2018 and received carbon ion radiotherapy. Positioning method: supine position with abdominal board and vacuum cushion molding fixation for the torso and both lower limbs, the affected limb elevated higher than the healthy limb, with no vacuum cushion obstruction within the irradiation field target area, and multiple marker lines drawn on the skin. Target delineation (Figure 2): the tumor bed and high-risk areas were determined based on two preoperative MRI and PET-CT images, with the Gross Tumor Volume (GTV) outlined as postoperative recurrent and residual lesions visible on CT and MRI, the Clinical Target Volume (CTV) expanded 1 cm axially and 5 mm in other directions from the GTV, appropriately retracted 3 mm away from the tibiofibular cortical bone, and the Planning Target Volume (PTV) expanded 3 mm from the CTV, with organs at risk including the tibia and fibula. Radiotherapy was performed using 2D uniform scanning technique with carbon ion beams at 260 MeV/u energy, featuring an 80 mm Bragg peak broadening, and a 100 mm HPDE compensator was added during treatment to optimize dose distribution, using bilateral opposing fields to irradiate the lesion, with a total dose of 64 Gy(RBE)/16 fractions (4.0 Gy(RBE)/fraction, 5 fractions per week). Dose Volume Histogram (DVH) analysis indicated that the 64.05 Gy(RBE) isodose curve encompassed 95% of the GTV, the 57.13 Gy(RBE) isodose curve encompassed 95% of the CTV, and the 49.62 Gy(RBE) isodose curve encompassed 95% of the PTV. The mean dose was 27.34 Gy(RBE) for the tibia and 44.27 Gy(RBE) for the fibula. Before each carbon ion radiotherapy session, positional verification was conducted, initially aligning according to skin marker lines, followed by adjustments based on bony landmarks under the carbon ion radiotherapy registration system iGPS by the attending physician, with this positioning method demonstrating stability and good reproducibility. Post-treatment regular follow-up examinations were performed, and treatment efficacy and adverse events were evaluated according to the RECIST 1.1 criteria for solid tumors and CTCAE 5.0, while radiation-induced injuries were assessed using the RTOG acute radiation injury grading scale.
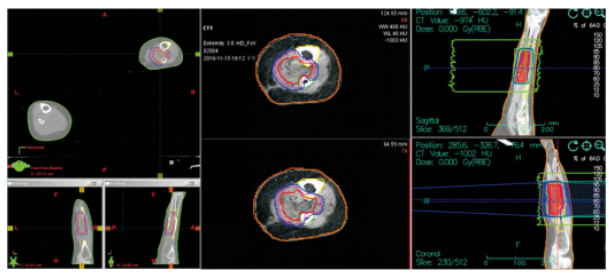
Fig.2 Target area delineation and treatment plan of the patient
Within 30 days after carbon ion therapy, the patient developed mild erythema and dry desquamation in the irradiated skin area, which was graded as Grade 1 radiation dermatitis according to the RTOG acute radiation injury scale and as Grade 1 radiation dermatitis according to CTCAE 5.0. Changes in skin reactions at 30, 60, and 90 days post-treatment are shown in Figure 3. No other treatment-related adverse reactions were observed. Treatment efficacy was evaluated based on enhanced MRI findings of the right lower limb, with specific results presented in Table 1 and Figure 4. The maximum diameter of the tumor lesion gradually decreased over the follow-up period. At 60 days after treatment completion, the sum of the diameters of the target lesions had decreased by 47.6% compared to the baseline, exceeding a 30% reduction, resulting in a PR (partial response) evaluation. By 120 days post-treatment, the target lesions had completely disappeared, leading to a CR (complete response) evaluation. At 270 days of follow-up, the efficacy evaluation remained CR (Figure 5), with no treatment-related late radiation injuries observed. No protocol deviations or violations occurred during the trial, and no serious adverse events (SAEs) were reported. The next phase will continue with the planned 5-year long-term follow-up.
2 Discussion
The standard treatment for bone and soft tissue sarcomas includes surgery and chemoradiotherapy. The role of neoadjuvant radiotherapy in reducing tumor size has been well established, particularly for soft tissue sarcoma patients, where it can lower local recurrence rates when combined with limb-sparing surgical treatment [6]. For extremity tumors, amputation may be a local treatment option, especially for tumors that are too large to resect while requiring preservation of neurovascular bundles. Except for small round cell sarcomas, conventional radiotherapy generally shows poor efficacy against bone and soft tissue malignancies [7]. Over the past three decades, the treatment approach for localized soft tissue sarcomas of the extremities has shifted from amputation and radical ablative surgery to more conservative, function-preserving surgery combined with radiotherapy [8-10]. McGee et al. [11] found that postoperative radiotherapy for extremity soft tissue sarcomas achieves good long-term disease control rates with acceptable toxicity.
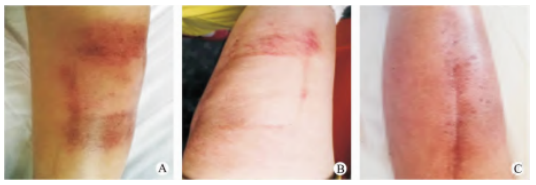
Fig.3 The skin reaction photos of the patient after treatment
A: 30 days after treatment. B: 60 days after treatment. C: 90 days after treatment.
Table 1. Efficacy Evaluation
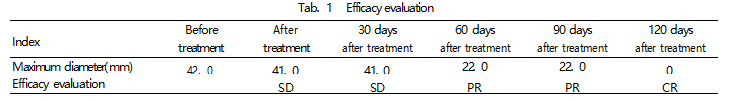
Note: SD: Stable disease. PR: Partial remission. CR: Complete remission.
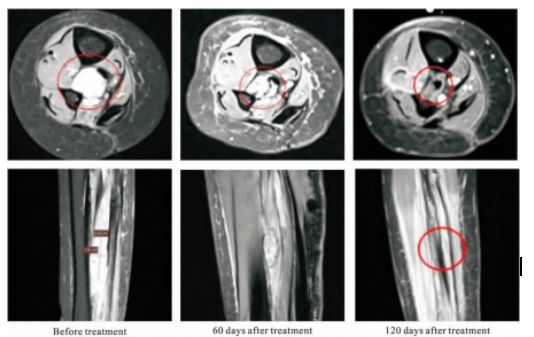
Fig.4 Comparison of MR examination results of the right lower limb before treatment, at 60 days after treatment ( PR) and at 120 days after treatment ( CR)
Sarcomas are radiation-resistant tumors, and conventional photon radiotherapy has poor efficacy. Carbon ion radiotherapy can induce more than two double-strand breaks in 70% of DNA molecules, with critical sites being more susceptible to damage and difficult to repair, leading to tumor cell death. Carbon ions kill tumor cells independently of the cell cycle. With a higher relative biological effectiveness (RBE), carbon ions demonstrate superior killing effects in necrotic and hypoxic tumor regions [12]. Additionally, carbon ion radiotherapy better spares normal tissues, exhibits improved tolerance to radiation therapy, produces fewer radiation-related toxic side effects, delivers stronger radiobiological effects against tumors, and shows enhanced efficacy against photon-radiotherapy-resistant tumors (including melanoma, osteosarcoma, chondrosarcoma, chordoma, and soft tissue sarcoma). Previous studies have reported on the efficacy of carbon ion therapy for bone and soft tissue sarcomas. SUGAHARA et al. [3] reported that carbon ion radiotherapy (CIRT) for primary localized sarcomas of the extremities achieved a 5-year overall survival rate of 56% and a 5-year local control rate of 76%, with no severe adverse reactions observed. In clinical trials conducted at the National Institute of Radiological Sciences in Japan, 128 cases of inoperable localized axial soft tissue sarcomas were treated with carbon ions, using a specific dose fractionation of 64.0-73.6 Gy(RBE)/(16f·4w). The resulting 3-year and 5-year local control rates were 68% and 65%, respectively, with 3-year and 5-year overall survival rates of 60% and 46%, and 3-year and 5-year disease-free survival rates of 39% and 32%. Only 4 patients experienced Grade 3 or higher late toxic side effects [13]. MATSUMOTO et al. [14] reported outcomes of carbon ion therapy for spinal sarcomas, with 5-year local control, overall survival, and progression-free rates of 79%, 52%, and 48%, respectively. Among 15 patients with tumor volumes less than 100 cm³, none experienced local recurrence. MOHAMAD et al. [15] reported 10 patients who survived more than 5 years (5-20.7 years) after carbon ion therapy, including 3-year and 5-year local control rates of 69.9% and 62.9% for inoperable pediatric osteosarcoma cases. Relevant research findings have also been reported in China. WEI Shihua et al. treated 3 cases of postoperative recurrent superficial soft tissue sarcomas using the heavy ion accelerator research facility at the Institute of Modern Physics, Chinese Academy of Sciences, achieving efficacy evaluations of CR in 2 cases and PR in 1 case after 3 months [16]. A retrospective study at Shanghai Proton and Heavy Ion Center on head and neck sarcoma patients with radiation or postoperative recurrence showed that proton or carbon ion therapy resulted in 1-year local control and overall survival rates of 74.6% and 86.5%, respectively, with only 2 cases of severe late adverse reactions [17]. For patients with inoperable bone and soft tissue sarcomas, treatment options for long-term prevention of local tumor progression are very limited. These findings represent highly encouraging results for such patients.
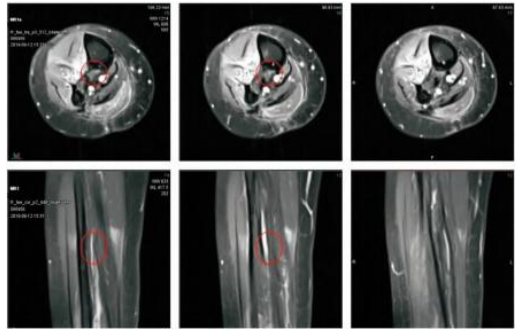
Fig.5 MR examination results ( CR) at 270 days after treatment
Heavy ion therapy emerged in the 1990s and has now become a frontier and hot topic in the global field of radiotherapy. The "Carbon Ion Therapy System" developed by the Institute of Modern Physics of the Chinese Academy of Sciences is the first domestically produced carbon ion therapy system with independent intellectual property rights in China. Installed at the Wuwei Cancer Hospital in Gansu Province, it has been officially approved as a Class III medical device product, registered, and formally put into clinical operation. Based on the aforementioned clinical results, carbon ion therapy has demonstrated good efficacy in treating photon radiation-resistant tumors such as bone and soft tissue sarcomas. During the clinical trial conducted from November 6, 2018, to May 23, 2019, to validate the efficacy and safety of China's independently developed carbon ion therapy system, one case of this type of tumor was included. The treatment achieved a PR evaluation at 2 months, a CR evaluation at 4 months, and remained a CR evaluation at 9 months. No adverse reactions above Grade II occurred during the treatment period or follow-up, indicating good short-term efficacy and minimal toxic side effects. This demonstrates that China's independently developed carbon ion therapy system is safe and effective for treating malignant tumors, but further follow-up is needed to observe long-term efficacy and to summarize treatment outcomes and adverse reaction results to better improve patient prognosis.
In summary, carbon ion therapy can achieve good efficacy and safety in treating bone and soft tissue sarcomas and may become a major and important non-invasive treatment method. However, many issues regarding carbon ion therapy still require further research, such as radiobiological effects, tumor-killing mechanisms and normal tissue damage, optimal dosage, and fractionation patterns. Additionally, the number of institutions currently offering carbon ion therapy and the number of patients receiving such treatment remain limited, necessitating further expansion.
【参考文献】
[1] JEMAL A,TIWARI RC,MURRAY T, et al. Cancer statistics[J]. CA: A Cancer Journal for Clinicians,2004,1( 1) : 8-29 .
[2] 中国抗癌协会肉瘤专业委员会,中国临床肿瘤学会.软组织肉瘤诊治中国专家共识( 2015 年版) [J].中华肿瘤杂志,2016,38 ( 4) : 310-320 .
Sarcoma Committee of Chinese Anti -Cancer Association, Chinese Society of Clinical Oncology. Expert consensus on diagnosis and treatment of soft tissue sarcoma ( 2015 edition) [J].Chinese Jour- nal of Oncology,2016,38( 4) : 310-320 .
[3] SUGAHARA S,KAMADA T,IMAI R , et al. Carbon ion radiotherapy for localized primary sarcoma of the extremities: Results of a phase I / II trial[J]. Radiotherapy and Oncology,2012,105 ( 2 ) : 226-231 .
[4] ITSUKO SERIZAWA,KENJI KAGEI,TADASHI KAMADA,et al. Carbon ion radiotherapy for unresectable retroperitoneal sarcomas [J]. International Journal of Radiation Oncology, Biology, Physics, 2009,75( 4) : 1105 -1110 .
[5] KEIJI MATSUMOTO,REIKO IMAI,TADASHI KAMADA, et al. Impact of carbon ion radiotherapy for primary spinal sarcoma [J]. Cancer,2013,119( 19) : 3496-3503 .
[6] STOJANINOVIC A,JAQUES DP,LEUNG DH, et al. Amputation for recurrent soft tissue sarcoma of the extremity: indication and
outcome [J]. Ann Surg Oncol,2001,8: 509.
[7] HAYASHI K,YAMAMOTO N,SHIRAI T, et al. Sequential histological findings and clinical response after carbon ion radiotherapy
for unresectable sarcoma [J]. Clinical and Translational Radiation Oncology,2017,2( Complete) : 41 - 45.
[8] PISTERS PW,O'SULLIVAN B,MAKI RG. Evidence - based recommendations for local therapy for soft tissue sarcomas[J]. J Clin Oncol,2007,25: 1003 - 1008.
[9] BACCI G,FERRARI S,BERTONI F, et al. Long - term outcome for patients with nonmetastatic osteosarcoma of the extremity treated at the Istituto Ortopedico Rizzoli according to the Istituto Ortopedico Rizzoli /osteosarcoma - 2 protocol: an updated report[J]. J Clin Oncol 2000,18: 4016 - 4027.
[10] HOEKSTRA HJ,SCHRAFFORDT KOOPS H,MOLENAAR WM, et al. A combination of intraarterial chemotherapy, preoperative
and postoperative radiotherapy, and surgery as limb - saving treatment of primarily unresectable high - grade soft tissue sarcomas of
the extremities[J]. Cancer,1989,63: 59 - 62.
[11] MCGEE L,INDELICATO DJ,DAGAN R, et al. Long - term results following postoperative radiotherapy for soft tissue sarcomas
of the extremity[J]. Int J Radiat Oncol Biol Phys,2012,84( 4) :1003 - 1009.
[12] KAMADA T,TSUJII H,BLAKELY EA, et al. Carbon ion radiotherapy in Japan: an assessment of 20 years of clinical experience
[J]. The Lancet Oncology,2015,16( 2) : e93.
[13] REIKO IMAI,TADASHI KAMADA,NOBUHITO ARAKI,et al.Carbon ion radiotherapy for unresectable localized axial soft tissue
sarcoma[J]. Cancer Medicine,2018,7( 9) : 4308 - 4314.
[14] MATSUMOTO K,IMAI R,KAMADA T,et al. Impact of carbon ion radiotherapy for primary spinal sarcoma[J]. Cancer,2013,
119( 19) : 3496 - 3503.
[15] MOHAMAD O,IMAI R,KAMADA T, et al. Carbon ion radiotherapy for inoperable pediatric osteosarcoma[J]. Oncotarget,2018,9 ( 33) : 22976 - 22985.
[16] 魏世华,李莎,张红,等. 12C6 + 重离子束照射治疗复发性软组织肉瘤临床疗效探讨[J]. 第四军医大学学报,2009,30( 5) :
451 - 453.WEI SH,LI S,ZHANG H,et al. Recrudescent soft tissue sarcoma treated by 12C6 + heavy ion beam rediotherapy[J]. J Fourth Mil Med Univ,2009,30( 5) : 451 - 453.
[17] 杨婧,王征,孔琳,等. 软组织肉瘤的放射治疗: 临床实践及进展[J]. 中国临床医学,2019,26( 3) : 326 - 331.YANG J,WANG Z,KONG L,et al. Radiotherapy for soft tissue sarcoma: clinical practice and progress[J]. Chinese Journal of Clinical Medicine,2019,26( 3) : 326 - 331.
Preliminary Review: Liu Wenyu
Final Review: Zhang Jie
