Carbon ion radiotherapy with complete tumor regression for primary malignant melanoma of female uret
Carbon ion radiotherapy with complete tumor regression for primary malignant melanoma of female urethra orifice: a case report
Xiaojun Li, Yihe Zhang, Yanshan Zhang, Yancheng Ye, Ying Qi, Tingchao Hu and Xin Pan
Abstract
Primary malignant melanoma of the female urethra (PMMFU) is extremely rare, accounting for 0.2% of all melanomas, and fewer than 200 cases have been reported worldwide. Because of the small number of clinical cases and unclear biological characteristics, there is no uniform and standard treatment protocol. We herein describe the treatment of PMMFU using carbon ion radiotherapy. The radiotherapy was delivered at 60.8Gy (RBE) in 16 fractions, once daily, five times per week. The patient achieved complete tumor disappearance within 1 year after carbon ion radiotherapy and remained disease-free thereafter. She developed acute grade 1 radiation dermatitis and urethritis, which resolved quickly; no other toxic effects were observed. At the time of this writing, her survival duration was 33 months. This case demonstrates that carbon ion radiotherapy may be a good option for primary genitourinary mucosal malignancies.
Introduction
Malignant melanoma (MM) is a highly malignant tumor of melanocytes that occurs in the skin, mucosa, and uvea.1 Mucosal MM (MMM) is a rare malignant tumor of mucosal origin, accounting for approximately 1% of all MMs.2 MMM occurring in the nasal cavity, oral cavity, and lip mucosa is rare; moreover, primary MM of the female urethra (PMMFU) is extremely rare,3 accounting for 0.2% of all melanomas.4 Because of the extremely small number of clinical cases of urethral MM and unclear biological characteristics, no consensus has been reached regarding the optimal treatment protocol. Most oncologists recommend radical resection, and classic radical surgery comprises partial vulvectomy and total urethrectomy (including resection of the anterior vaginal wall and bladder neck) with inguinal lymph node dissection. Additional procedures may include pelvic lymph node dissection, reconstruction of the urethra using a bladder muscle flap, and even anterior hemipelvic radical surgery.5 However, such invasive treatments deprive patients of their normal functions.Nevertheless, management of PMMFU recurrence remains challenging, and a local recurrence rate of up to 60% to 70% at 1 year after surgery has been reported.6
Radiotherapy is an effective local treatment for patients with inoperable tumors and those who are reluctant to undergo surgery. However, MMM is usually is an X-ray-resistant tumor.7 Carbon ion radio-therapy (CIRT) has distinct physical characteristics, including a sharp lateral penumbra, very low energy deposition within the entry path prior to the Bragg peak formed by the steep dose deposition, and a sharp dose fall-off after the Bragg peak, thus facilitating dose delivery within a finite range.8 In addition to its superior physical properties, CIRT is a modality with high linear energy transfer, and the relative biological effectiveness (RBE) of CIRT is significantly higher than that of photon and proton radiation. The RBE value is 2 to 5 for CIRT depending on the tissue type and endpoint of study. It has been suggested that more damage from radiation with high linear energy transfer occurs in the form of direct DNA double-strand breaks, which is more difficult to repair.9 These properties together are beneficial in treating tumors that are not sensitive to X-ray radiation. CIRT for urothelial melanoma has rarely been reported. We herein describe a patient with PMMFU who underwent CIRT, achieved complete tumor disappearance within 1 year after completion of CIRT,and remained disease-free thereafter; at the time of this writing, her survival period was 33 months. This case is reported in accordance with the CARE guidelines.10
Case presentation
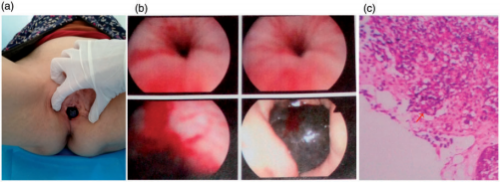
Figure 1. (a) Malignant melanoma of the urethral orifice was observed. (b) Cystoscopy showed a dark brown mass of about 3cm in the urethral orifice. (c) Microphotograph (×100) from a fine needle aspiration biopsy specimen showed characteristic malignant melanoma cells.
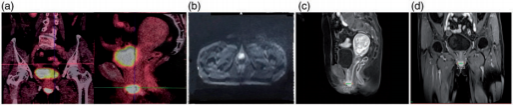
Figure 2. Images of the urethral orifice melanoma. (a) Positron emission tomography/computed tomography imaging. Before treatment, a high standardized uptake value was observed. (b) Diffusion-weighted imaging demonstrated a high-intensity lesion. (c) T1-weighted contrast-enhanced sagittal imaging also demonstrated a high-intensity lesion. (d) Coronal magnetic resonance imaging.
A woman in her early 70s was referred to our center to undergo CIRT for a urethral orifice MM. In the latter half of 2018, she had experienced discomfort and a swelling sensation of the urethral orifice while performing physical activity, and these symptoms were accompanied by urinary frequency and urgency. The symptoms gradually became aggravated during the next 3 months. Soon thereafter, a nontender, dark brown, indurated mass resembling a plum was found at the urethral orifice (Figure 1(a)). During the following 3 months, the mass progressively enlarged and the patient developed dysuria and a burning sensation during urination. At the end of 2018, cystoscopy at a local hospital showed an approximately 3-cm dark brown mass at the urethral orifice (Figure 1(b)). Pathological examination of a needle biopsy specimen showed MM, and immunohistochemical examination showed CKP (-), HMB45 (-), S-100 (-), melan-A (+), vimentin (+), CD30 (-), and Ki-67 (+70%) (Figure 1(c)). Fluorodeoxyglucose-positron emission tomography/computed tomography (CT) showed abnormally increased fluorodeoxyglucose uptake in the urethral orifice (29 × 26 × 26mm) with a maximum standardized uptake value of 48.6 (Figure 2(a)).
Magnetic resonance imaging (MRI) showed a round high-intensity lesion at the urethral orifice on T2-weighted imaging, similar to the findings on T1-weighted imaging and diffusion-weighted imaging (Figure 2(b–d)), with an apparent diffusion coefficient of 0.88×10⁻³ mm²/s and significant enhancement (Figure 2(c) and (d)). On gynecologic examination, a round mass was found at the urethral orifice at the 6-o’clock position with the patient in the lithotomy position. The lesion invaded the vaginal wall, measured approximately 2.5 × 2.4 × 2.0cm, and was dark brown and hard on palpation. It had a smooth surface and limited mobility, no tenderness, no obvious ulcer or bleeding, and no purulent secretion. The diagnosis was urethral MM cT4aN0M0 IIb (Revised American Joint Committee on Cancer 2002 tumor-node-metastasis melanoma staging system).¹¹ The patient had no cancer-related family history. She refused to undergo surgical treatment, so she was transferred to our center for CIRT. The patient was immobilized in the lithotomy position and underwent CT simulation using a four-dimensional CT system with 3-mm-thick slices. Treatment planning was performed using the ciPlan system, version 1.0 (Institute of Modern Physics, Lanzhou, China). The target area was delineated on the CT images. The gross target volume was the urethral orifice mass on CT, fused with the MRI and positron emission tomography/CT images. The clinical target volume included the gross target volume plus a 5-mm margin to encompass the surrounding tissue at risk of microscopic involvement. The planning target volume consisted of the clinical target volume plus an isotropic 3-mm margin for positioning errors during daily radiotherapy.
A passive scattered carbon ion beam (Heavy Ion Medical Machine; Lanzhou Kejin Taiji Corporation, Ltd.) was used for the treatment. CIRT planning was performed using biologic treatment plan optimization by the ciPlan treatment planning software system, which takes into account local values of the RBE calculated by the ciPlan software based on the mixed beam model. The mixed beam model was developed to predict the RBE of the passively scattered carbon ion beams with the tumor response as the relevant endpoint. The calculated radiation dose for the target volume and surrounding normal structures was expressed as the Gy RBE-weighted dose, which was defined as the physical dose multiplied by the carbon ion RBE, which is approximately 3.0 at 0.8cm from the distal end of the spread-out Bragg peak.
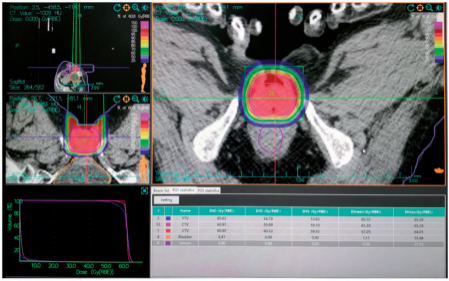
Figure 3. Carbon ion radiotherapy target and dose distribution.
CIRT was delivered at a 60.8-Gy RBE-weighted dose in 16 fractions with a 190-MeV/U carbon ion beam, once daily, five times per week (Monday to Friday) for 22 days using one vertical portal (Figure 3).
Efficacy was evaluated according to the RECIST 1.1 criteria.12 Acute reactions of normal tissue were classified according to the Cancer Therapy Evaluation Program, Common Terminology Criteria for Adverse Events, Version 4.0,13 with a maximum reaction occurring within 3 months after initiation of therapy. Late reactions were classified according to the Radiation Therapy Oncology Group/European Organization for Research and Treatment of Cancer scoring system.14
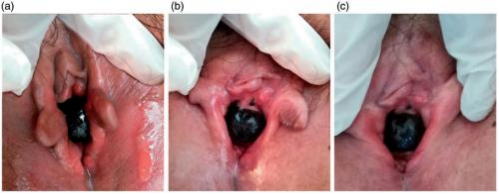
Figure 4. Skin and mucosa reactions at 10, 30, and 60 days after the start of carbon ion radiotherapy.
Table 1 CIRT, Evaluation of efficacy according to maximum diameter of tumor.
| After CIRT | ||||||
| Time | Before CIRT | End of CIRT | Day 30 | Day 90 | Day 180 | 1 year |
| Diameter, mm | 25 | 26 | 23 | 17 | 12 | 0 |
| Tumor response | – | SD | SD | PR | PR | CR |
CIRT, carbon ion radiotherapy; SD, stable disease; PR, partial response; CR, complete response.
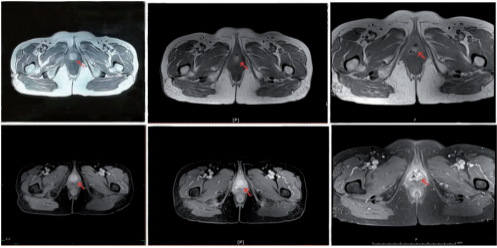
Figure 5. Comparison of imaging data before carbon ion radiotherapy, 90 days after carbon ion radiotherapy (partial response), and 180 days after carbon ion radiotherapy (partial response).
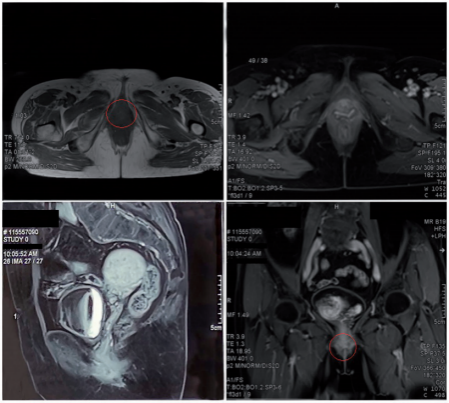
Figure 6. Gadolinium-enhanced magnetic resonance imaging 1 year after carbon ion radiotherapy showed that the lesion had completely disappeared.
During CIRT, dysuria and frequency (up to 9–10 times/day) occurred from the seventh fraction of treatment. These symptoms were considered to have been caused by radiation-induced urethral edema, and they were relieved by symptomatic treat ment. Skin and mucosal erythema in the irradiated field occurred 10 days after the start of CIRT, as did edema of the labia major and minor, the vaginal opening, and the urethral orifice around the lesion; these were assessed as grade 1 radiation dermatitis and mucositis. The skin and mucosal adverse effects were relieved 1 month after completion of CIRT, and the dermatitis, mucositis, dysuria, and frequency of urination were totally resolved 2 months after completion of CIRT (Figure 4). No other acute or chronic adverse effects of CIRT were observed. At the end of CIRT, the tumor treatment response was stable disease. Three months after completion of CIRT, the lesion was visually smaller than before, and MRI showed that the maximum tumor diameter was about 17mm with a 32% reduction (partial remission). The lesion completely disappeared 1 year after treatment (Table 1, Figure 5, and Figure 6). The patient declined other systemic treatment after CIRT for personal reasons. As of November 2021, 33 months after CIRT, the patient was alive and disease-free. She was satisfied with the outcome of CIRT. No severe reactions were observed.
Discussion
PMMFU accounts for 0.2% of all melanomas and 4% of all urothelial carcinomas. Fewer than 200 cases have been reported in the literature.15,16 About 50% of mucosal melanomas occur in the head and neck region (nasal cavity, sinuses, and oropharynx), about 30% occur in the gastrointestinal tract (esophagus, rectum, and analcanal), and approximately 20% involve the genitourinary tract (vagina, cervix, bladder, and urethra); mucosal melanomas occasionally originate from other mucosal areas.17 Clinical manifestations of PMMFU include a mass of the urethra, hematuria, and urinary tract obstruction. Our patient presented with a mass in the external urethral orifice with a typical dark brown appearance.
Urethral tumors are among the rarest types of cancer in the female urogenital tract. Cystourethroscopy permits visualiza- tion of urethral tumors and facilitates biop- sies for histological examination. MRI is recommended for tumor staging. Local, superficial, and distal urethral tumors may be treated by partial resection with preservation of the urethra. Radical urethrectomy and wide resection of the paraurethral tissues and anterior vaginal wall may offer superior local control of this disease. However, advanced female urethral tumors and lymph node positivity are associated with a poor prognosis for all survival outcomes (recurrence-free, cancer-specific, and overall survival). Multimodal therapy including surgery, chemotherapy, and radiotherapy are required in modern management regimens, although the specific role and combination of each treatment is less clearly determined. Wide resection after chemotherapy and/or radiotherapy is asso- ciated with the best local control, but it reduces the patient’s quality of life.18
There is no standard treatment protocol for urothelial MM, but most experts recommend radical resection.17 However, because of the complex anatomy and delicate phys- iological function of the urogenital tract in which the tumor is located, achieving R0 resection with surgical treatment is often challenging and is likely to result in the loss of physiological function. Furthermore, the recurrence rate of urothelial MM is as high as 71% regardless of the surgical approach.17 Davuluri et al.6 described a patient with a 49-mm PMMFU who developed recurrence and metastasis 5 months after surgical treatment. The patient underwent salvage pelvic radiotherapy and immunotherapy and eventually died of metastatic disease 11 months after surgery.6 Broussard et al.19 described an 87-year-old woman with urethral MM who underwent extensive local tumor resection and achieved postoperative survival of only 3 months.
Radiation therapy is a widely used and effective local treatment for many malignant tumors, and it is often the only alternative local treatment available for patients not amenable to surgery. However, MMM is resistant to X-ray radiation. The radiation-resistant nature of melanoma results in limited efficacy of conventional radiation in the treatment of MMM. CIRT is a highly precise radiation therapy technique because of its physical properties. It also provides higher RBE that can be used for the treatment of malignancies resistant to photon radiation. In addition, early results of clinical studies have suggested that such advantages might translate into improved clinical outcomes.20 CIRT exhibits improved treatment outcomes in tumors resistant to photon radiotherapy including melanoma, osteosarcoma, chondrosarcoma, chordoma, and other soft tissue sarcomas.21
A systematic review of the effectiveness and safety of CIRT for MM reported pooled data from 11 studies, showing that patients with MM treated with CIRT exhibited better local control, low recur- rence rates, and mild toxicity at sites includ- ing the skin, mucosa, and choroid.22 Murata et al.23 examined the long-term treatment outcomes of CIRT for gyneco- logic MM with tumor sites in the vagina vulva, and uterus and observed complete tumor disappearance in 30 of 37 patients (81%); the 2-year local control, overall survival, and progression-free survival rates were 71%, 53%, and 29%, respectively. These results are encouraging for MM in the genitourinary tract and conform to our experience in this case report. Because PMMFU is very rare, there is no standard protocol for CIRT, and very few reports have described prophylactic irradiation of lymph nodes using CIRT. Therefore, our multidisciplinary team decided to perform CIRT only for the primary tumor; regional lymph node irradiation was not performed.
Wuwei Heavy Ion Center is a carbon ion facility in China dedicated to medical use and was designed by the Institute of Modern Physics of the Chinese Academy of Sciences. We used this facility to treat tumors since November 2018.24 Few reports worldwide have described patients who underwent CIRT for treatment of PMMFU. In the present case, we treated one patient with CIRT and achieved a good outcome with no adverse events. At the time of this writing, the patient was disease-free with a survival duration of 33 months after CIRT.
Our report demonstrates that complete tumor regression is possible after CIRT in patients with PMMFU. Longer follow-up and larger numbers of patients are needed to further assess the long-term effectiveness and late toxicity-related adverse effects of this treatment.
Ethics statements
This study was approved by the ethics review committee of Wuwei Cancer Hospital. Written informed consent was obtained from the patient.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was supported by the Key R&D Plan of Science and Technology Program of Gansu Province, China (no. 19YF3FH001).
ORCID iD
Yanshan Zhang https://orcid.org/0000-0003- 4621-148X
References
1. Kibbi N, Kluger H, and Choi JN. Melanoma: clinical presentations. Cancer Treat Res 2016; 167: 107–129.
2. Jawad MB and Kofoed K. Mucosal malig- nant melanomas in the gastrointestinal tract and urogenital organs. Ugeskr Laeger 2017; 179: V09160679.
3. Barnhill RL and Gupta K. Unusual variants of malignant melanoma. Clin Dermatol 2009; 27: 564–587.
4. Safadi A, Schwalb S, Ben-Shachar I, et al. Primary malignant urethral melanoma resembling a urethral caruncle. Urol Case Rep 2017; 15: 28–29.
5. Todo Y, Okamoto K, Suzuki Y, et al. Radicality of initial surgery for primary malignant melanoma of the vagina. Melanoma Res 2016; 26: 173–180.
6. Davuluri M, Long B, Semple S, et al. Primary urethral melanoma: a case report and literature review. Urology 2019; 126: 1–4.
7. Qin J, Li S, Zhang C, et al. Apoptosis and injuries of heavy ion beam and x-ray radia- tion on malignant melanoma cell. Exp Biol Med (Maywood) 2017; 242: 953–960.
8. Lin R, Slater JD, Yonemoto LT, et al. Nasopharyngeal carcinoma: repeat treat- ment with conformal proton therapy— dose-volume histogram analysis. Radiology 1999; 213: 489–494.
9. Huang YW, Pan CY, Hsiao YY, et al. Monte Carlo simulations of the relative bio- logical effectiveness for DNA double strand
breaks from 300MeVu1 carbon-ion beams. Phys Med Biol 2015; 60: 5995–6012.
10. GagnierJJ, Kienle G, Altman DG, et al. The CARE guidelines: consensus-based clinical case reporting guideline development. Headache 2013; 53: 1541–1547.
11. Piura B. Management of primary melanoma of the female urogenital tract. Lancet Oncol 2008; 9: 973–981.
12. Eisenhauer EA, Therasse P, BogaertsJ, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009; 45: 228–247.
13. NCI Common Terminology Criteria for Adverse Events (CTCAE) V4.0 Data Files. Available online: http: //evs.nci.nih.gov/ ftp1/CTCAE/About.html (accessed 15 January 2019).
14. Cox JD, Stetz J and Pajak TF. Toxicity cri- teria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys 1995; 31: 1341–1346.
15. Walsh PC, Retik AB, McDougal WC, et al. Surgery of penile and urethral carcinoma. In: Wein AJ, Kavoussi LR, Novick AC, et al. (eds) Campbell-Walsh Urology. 10th ed. Philadelphia: WB Saunders, 2012. pp.946–951.
16. El-Safadi S, Estel R, Mayser P, et al. Primary malignant melanoma of the ure- thra: a systematic analysis of the current lit- erature. Arch Gynecol Obstet 2014; 289: 935–943.
17. Kalogirou EM, Kalyvas D, Tosios KI, et al. Recurrence in a patient with a 10-year his- tory of sinonasal mucosal melanoma mani- festing as facial swelling. J Clin Exp Dent 2017; 9: e1492–e1495.
18. Dell’Atti L and Galosi AB. Female urethra adenocarcinoma. Clin Genitourin Cancer 2018; 16: e263–e267. doi: 10.1016/j. clgc.2017.10.006
19. Broussard AP, Chaudoir C and Gomelsky A. Urethral melanoma in an elderly woman. Int Urogynecol J 2015; 26: 149–150.
20. Habermehl D, Wagner M, Ellerbrock M, et al. Reirradiation using carbon ions in patients with locally recurrent rectal cancer at HIT: first results. Ann Surg Oncol 2015; 22: 2068–2074.
21. Kamada T, Tsujii H, Blakely EA, et al. Policy Review Carbon ion radiotherapy in Japan: an assessment of 20 years of clinical experience. Lancet Oncol 2015; 16: e93–e100.
22. Li C, Zhang Q, Li Z, et al. Efficacy and safety of carbon-on radiotherapy for the malignant melanoma: A systematic review. Cancer Med 2020; 9: 5293–5305.
23. Murata H, Okonogi N, Wakatsuki M, et al. Long-term outcomes of carbon-ion radio- therapy for malignant gynecological mela- noma. Cancers (Basel) 2019; 11: 482.
24. Li X, Zhang Y, Ye Y, et al. Carbon ion radiotherapy for recurrent calf myxoid lip- osarcoma: a case report. J Int Med Res 2021; 49: 3000605211009701.
初审:张莉红 复审:张洁
