Heavy Ion Technology Ushers in New Era for Benign Disease Treatment
Heavy Ion Technology Ushers in New Era for Benign Disease Treatment
Low-dose heavy ion therapy for refractory benign diseases such as rheumatoid arthritis has begun clinical research and treatment at Gansu Wuwei Cancer Hospital. The cost of heavy ion therapy is covered by research project funding, with zero out-of-pocket expenses for patients.
Rheumatoid arthritis (RA) is an autoimmune disease characterized by chronic synovitis, progressive joint destruction, and systemic inflammation, with a global prevalence of approximately 0.5%–1%. It severely impacts patients' quality of life and imposes a significant socioeconomic burden. Although the use of biologics (e.g., TNF-α inhibitors, IL-6 receptor antagonists) and small molecule targeted drugs (e.g., JAK inhibitors) has significantly improved the prognosis of RA, about 30%–40% of patients respond poorly to or cannot tolerate existing treatments, and some develop complications such as severe infections. Therefore, there is an urgent need to explore new therapeutic strategies.
Low-dose radiotherapy (LDRT), typically defined as a single dose ≤1 Gy and total dose ≤6 Gy, has a history of use in treating RA dating back to the early 20th century. In the 1970s, it was considered one of the most effective physical therapies for spinal and joint diseases. However, with the rise of DMARDs, LDRT gradually faded from mainstream treatment. In recent years, advances in radiobiological research have gradually elucidated the anti-inflammatory and immunomodulatory mechanisms of LDRT, providing a theoretical basis for its modern application. At the same time, the development of advanced radiotherapy technologies such as heavy ion therapy offers new possibilities for treating refractory RA.
Supported by the Chief Scientist Project of Gansu Province titled "Research on Heavy Ion Therapy for Non-Tumor Diseases," and with the backing of a research team led by the Institute of Modern Physics, Chinese Academy of Sciences, Gansu Wuwei Tumor Hospital and affiliated research groups have officially launched research on heavy ion therapy for rheumatoid arthritis this year. This study aims to overcome the limitations of traditional treatments and provide an innovative "Chinese solution" for patients with refractory rheumatoid arthritis. At the same time, the research will promote the expansion of heavy ion medicine into the field of non-tumor diseases, offering new treatment options and hope for recovery to RA patients worldwide.

Inclusion Criteria for Volunteers
- Voluntarily participate in the clinical study, fully understand the study content, provide informed consent, and take a key step in advancing medical science of their own free will.
- Age ≥ 30 years;
- Diagnostic criteria: Meet the 2010 ACR/EULAR classification criteria for RA; disease duration ≥ 6 months;
- Disease activity: DAS28-CRP > 3.2 (moderate to high activity), with at least one joint showing ultrasound-confirmed synovitis (PD signal ≥ grade 2);
- Refractory definition: Poor response (DAS28 improvement < 1.2) after treatment with ≥ 2 biologic DMARDs (including at least one TNF-α inhibitor) for ≥ 3 months.The above criteria are the main inclusion criteria. Final eligibility for enrollment will be determined by the investigator in accordance with the study protocol.
Volunteer Exclusion Criteria
- Patients with a history or current presence of severe dysfunction of the heart, lungs, liver, kidneys, or other vital organs;
- Previous radiotherapy (including LDRT) to any joint area;
- Patients with allergic reactions or contraindications to radiotherapy;
- Other infectious diseases;
- Currently participating in another clinical study, or less than 30 days have passed since the completion of radiotherapy;
- Severe joint deformity requiring surgical intervention (e.g., indications for joint replacement);
- Pregnant or breastfeeding women (women of childbearing age must use contraception);
- Occurrence of a cardiovascular or cerebrovascular event within the past 6 months;
- Concurrent other autoimmune diseases (e.g., SLE, scleroderma).
Experimental Design
- Strictly adhere to the volunteer recruitment principles for clinical trials, fully respect patients' personal treatment preferences, and divide enrolled volunteers into three low-dose heavy ion irradiation groups with total doses of 2 Gy, 3 Gy, and 4 Gy, respectively. Using the advanced heavy ion accelerator at our hospital, low-dose irradiation interventions will be administered to patients accordingly.
- During the key periods before and after low-dose radiotherapy, a comprehensive series of assessments will be conducted, including knee joint imaging and pathology evaluation, joint effusion specimen collection, synovial biopsy, blood specimen collection, pain assessment, functional assessment, safety evaluation, adverse event evaluation, and efficacy assessment. Through rigorous pre- and post-treatment comparative analysis, the clinical effectiveness of low-dose radiotherapy will be precisely determined.
- Pre-treatment screening evaluation is critically important. After providing informed consent (ICF), eligible patients will immediately enter the screening process, during which detailed disease-related information will be collected and recorded, and comprehensive examinations will be arranged. Based on the latest examination results, investigators will rigorously determine whether patients meet the inclusion and exclusion criteria.
- The follow-up period will begin immediately after treatment completion. From the end of the last study treatment, all subjects will enter a 1-year safety follow-up period, during which regular safety assessments and adverse event monitoring will be conducted. Follow-up time points are set as: before treatment, and at 3 weeks, 6 weeks, 9 weeks, 12 weeks, 6 months, and 1 year after treatment initiation. Additionally, all adverse events potentially related to the study treatment occurring from the time of ICF signing through 30 days after the last treatment (including day 30) will be comprehensively collected and scientifically evaluated based on incidence and severity.
Patient Rights
- The hospital will provide research funding to the expert team to cover patients' heavy ion therapy costs.
- A multidisciplinary expert team (MDT) will develop personalized treatment plans throughout the entire process.
- Patients will receive regular follow-up monitoring within three years after treatment and will have priority access to new therapies.
Contact Person(+86-)
Su Anping: 13893592413
Yang Qianzi: 15379332290
Ma Haitao: 17793510936
Xu Jianping: 18740983170
Wang Haiqing: 15193560091
Yin Lirong: 17339942775
Gansu Wuwei Cancer Hospital Wuwei Heavy Ion Center




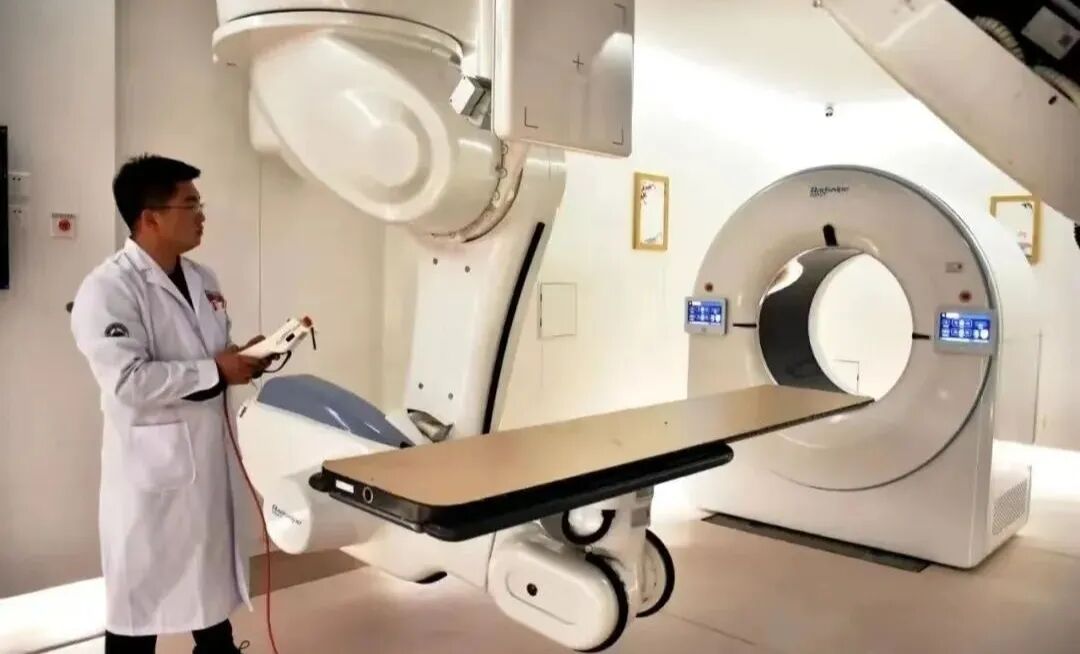
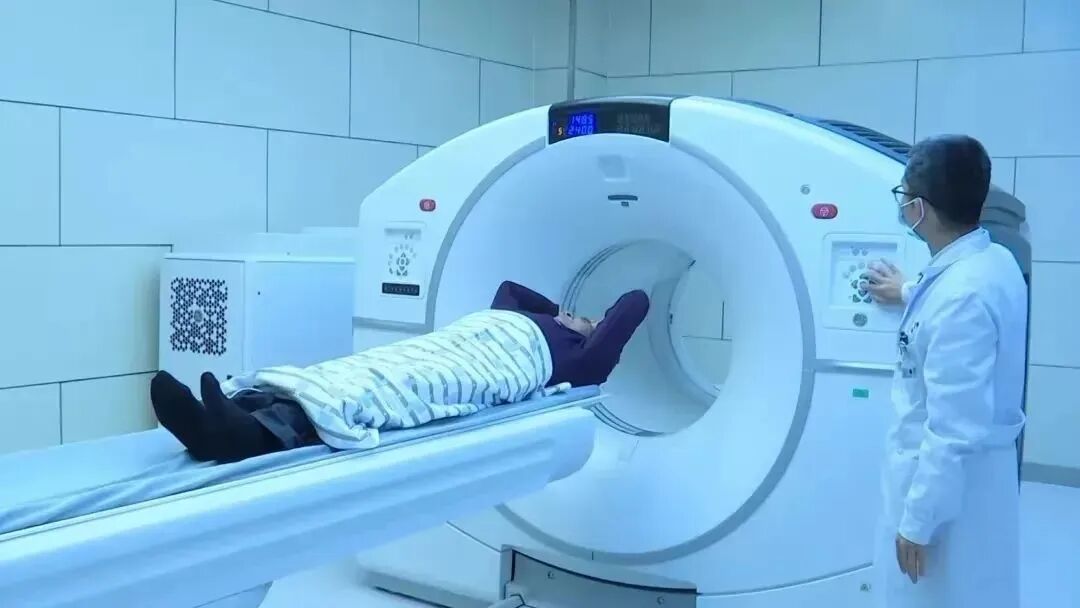
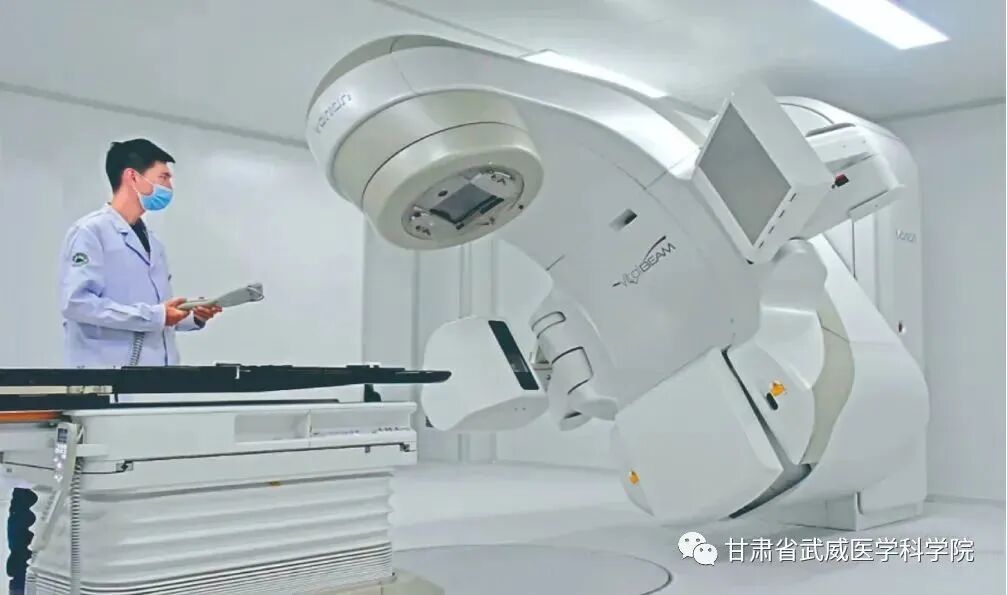
Gansu Wuwei Cancer Hospital (Wuwei Institute of Medical Sciences)
(Gan) Medical Advertisement [2025] No. 11-14-387
